Artur Palasz, Ph.D., SPEKTROCHEM – Formulation and Raw Materials Research Laboratory for Architectural Paints, Poland; e-mail: artur.palasz@spektrochem.pl03.22.22
INTRODUCTION
Mineral thickeners are a group of organoclays that occur naturally in the form of deposits. They are made of laminar plates and are phyllosilicates used in a wide range of industries, from sorbents to cat litter. Due to their high ability to absorb liquids and form gels, they are also used as paint thickeners. Naturally occurring and modified bentonites, attapulgites, smectites and layered aluminosilicates are of particular importance.
Mineral thickeners are a very important group of rheological modifiers of architectural paints. They are responsible for the rheological properties of latex paints in the low (Brookfield-viscosity) and mid-shear (Stormer-viscosity) areas and for the synergy of action with rheology modifying thickeners in the areas of high shear forces (ICI-viscosity). In the area of low and mid-shear rates, they are responsible for imparting viscosity and maintaining the rheological properties of sagging, settling and sedimentation, storage stability, including syneresis prevention and the “feeling” of in-can viscosity. They work very well with other thickeners responsible for rheology in the area of low and medium shear rates, incl. cellulose derivatives and high-shear ICI-builders in elimination
of spattering.
Their ability to absorb liquids and form gels is closely related to their structure and the possibility of changes that occur during their swelling in the water environment. But is this swelling the same in all water? After all, as you know, water-based paints are made of both demineralized and tap water. Different producers also use post-process water (waste-water, recycled water), and the tap water has a different hardness, which in turn is related to different ions in its composition. This article discusses the basic influence of the type of water on the hydration of mineral thickeners, as well as their influence on the properties of paints prepared from them. I was inspired to write this article by the story of one of the paint manufacturers who, after opening a latex paint factory in another part of the country and despite using the same raw materials, could not obtain the same viscosity from the same formulation as in his other factory.
HYDRATION AND HOUSE OF CARDS
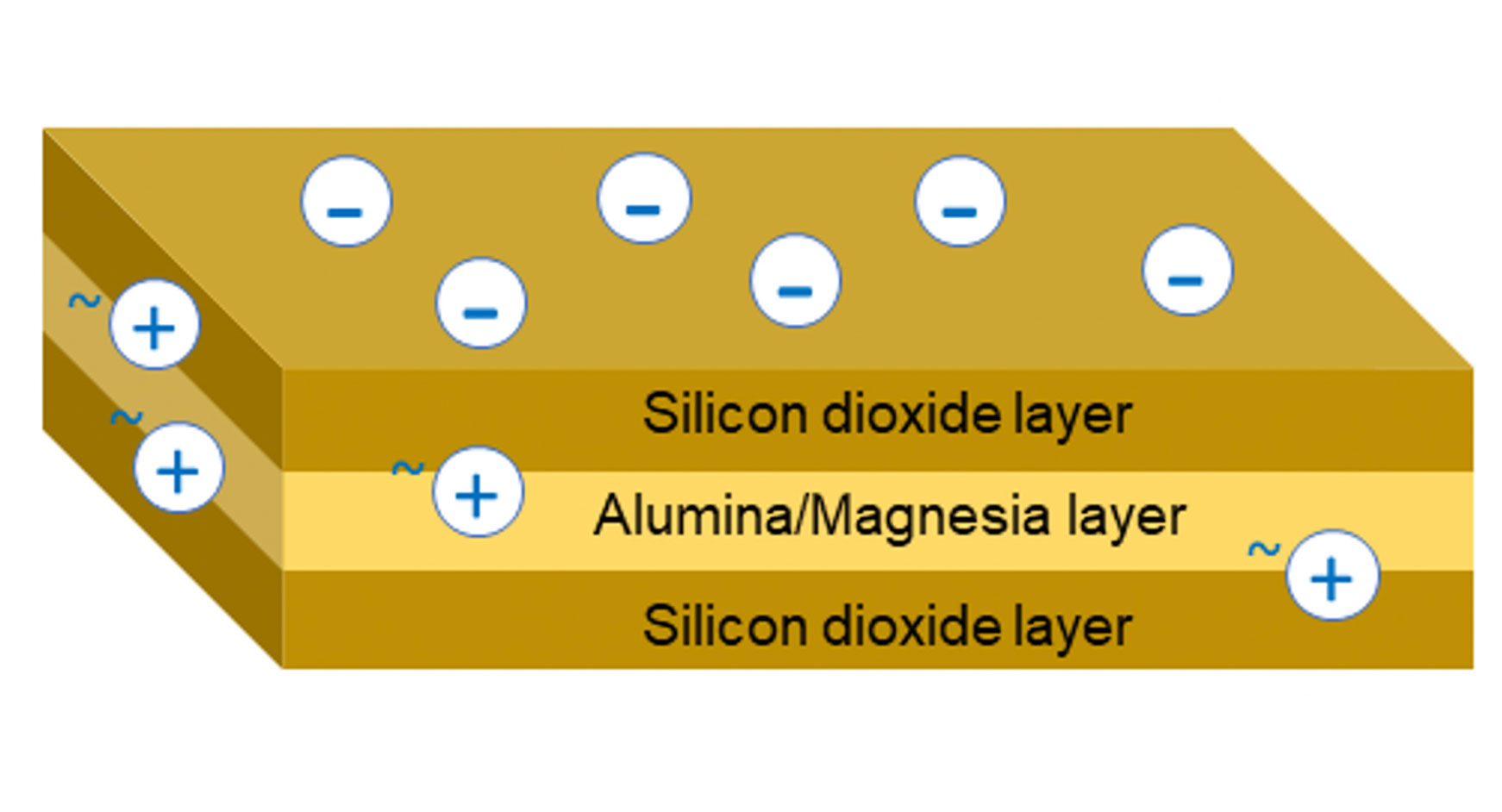
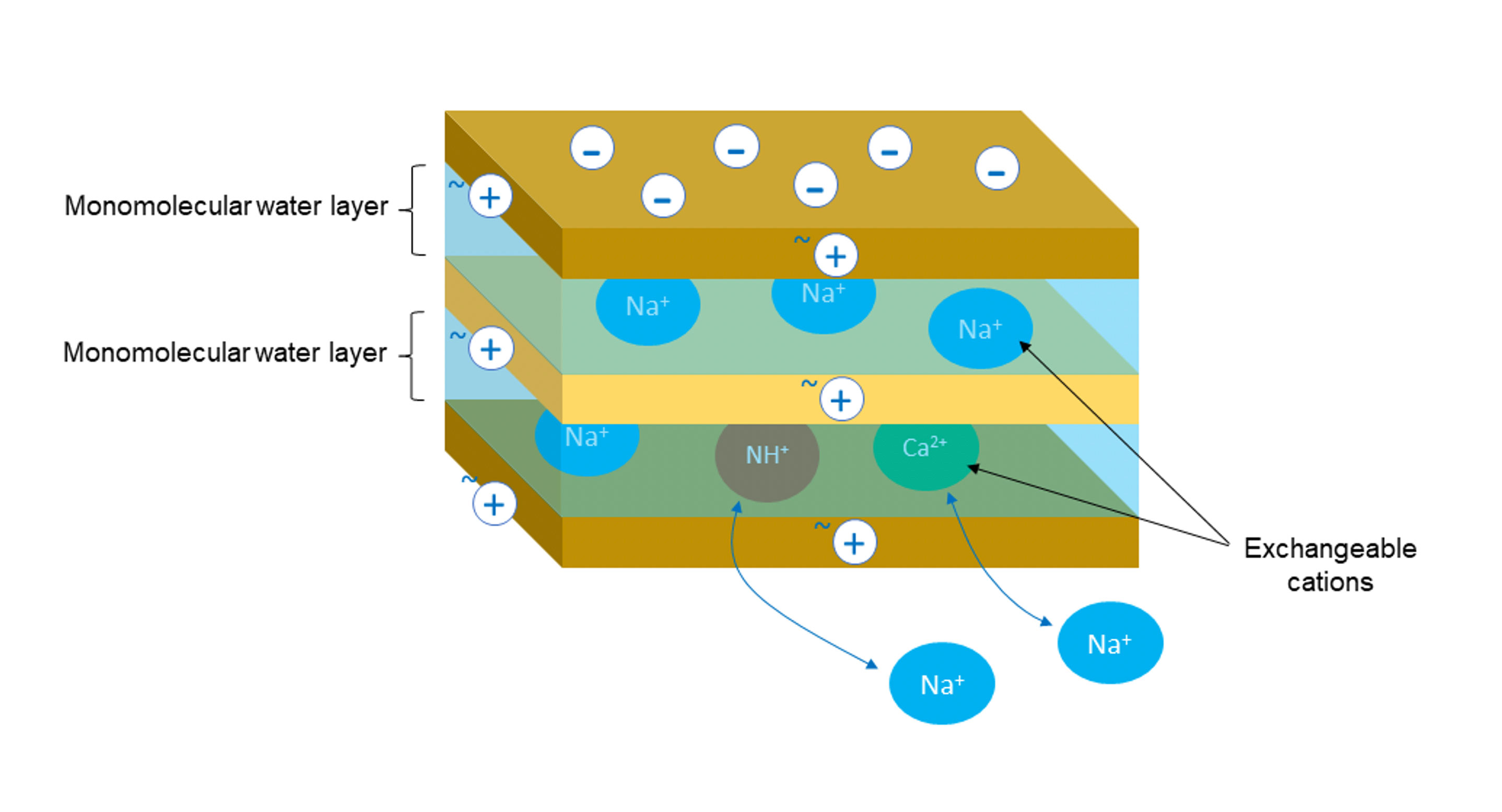
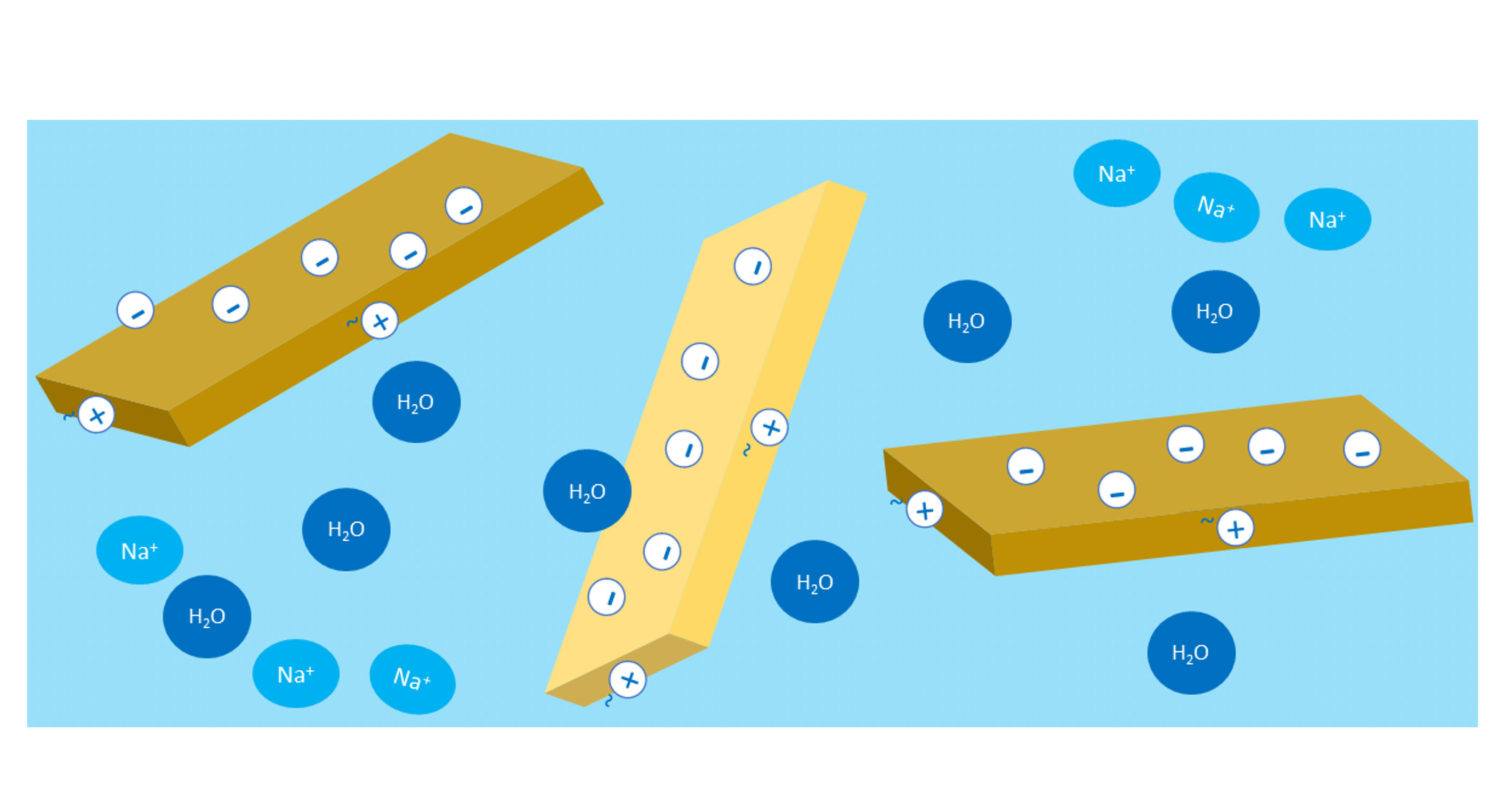
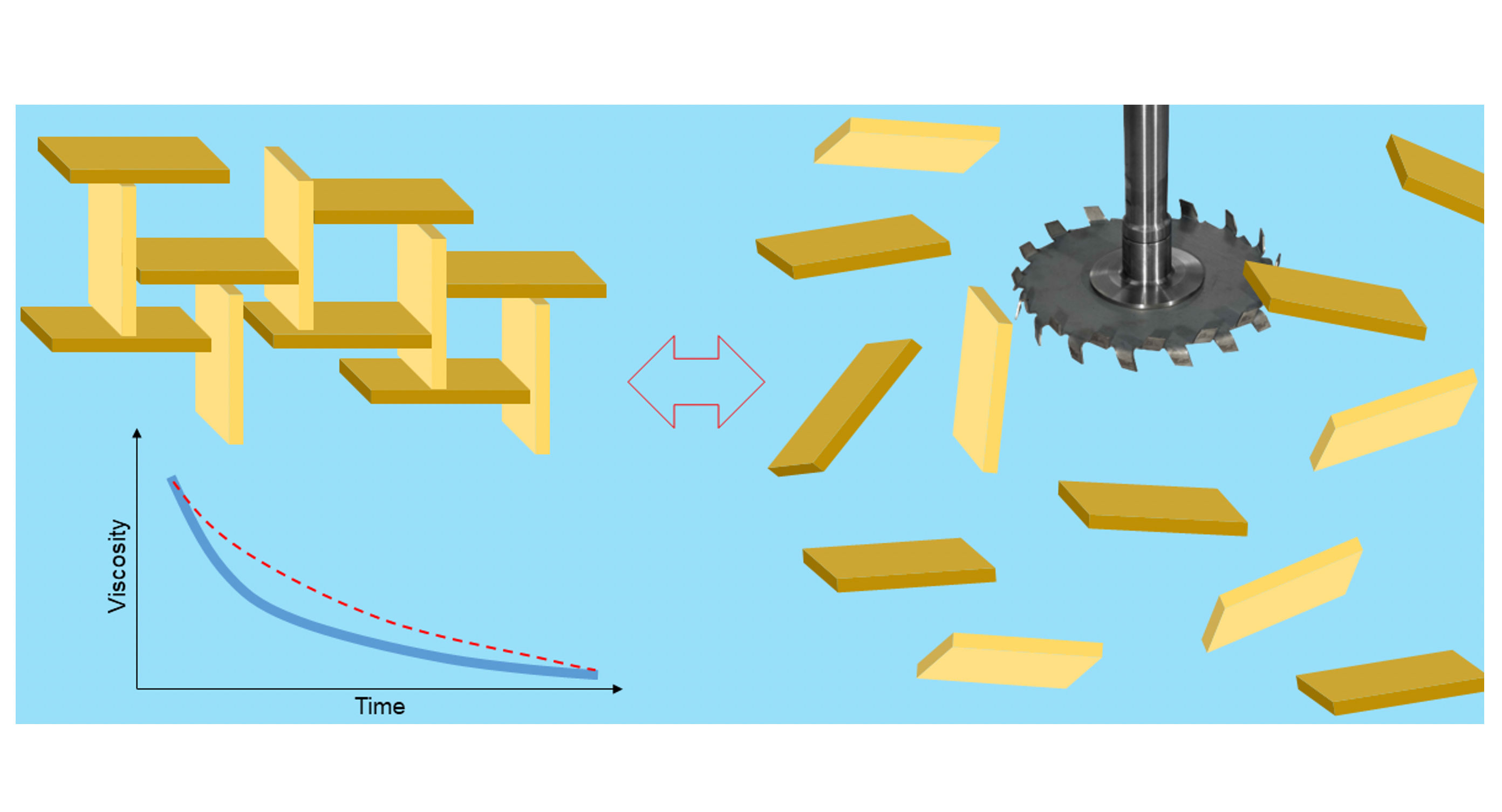
Mineral thickeners, e.g. smectites, are made of layers of platelets with dimensions of approximately 100 x 100 nm and a thickness of approximately 1 nm (0.001 µm). Their edges are slightly positively charged, while their platelet faces are negatively charged [Figure 1]. The negative surface charge is balanced by sodium ions, which are exchangeable and can be substituted by other cations, e.g. NH+, Ca2+ [Figure 2]. A single grain of clay is made up of thousands of sandwich platelet with exchangeable ions and a monomolecular water layer in between each. When the clay is dispersed in the water, the water penetrates the spaces between the platelets. Then ions are exchanged and replaced with water molecules (diffusion and osmosis), which results in delamination of the plates [Figure 3].
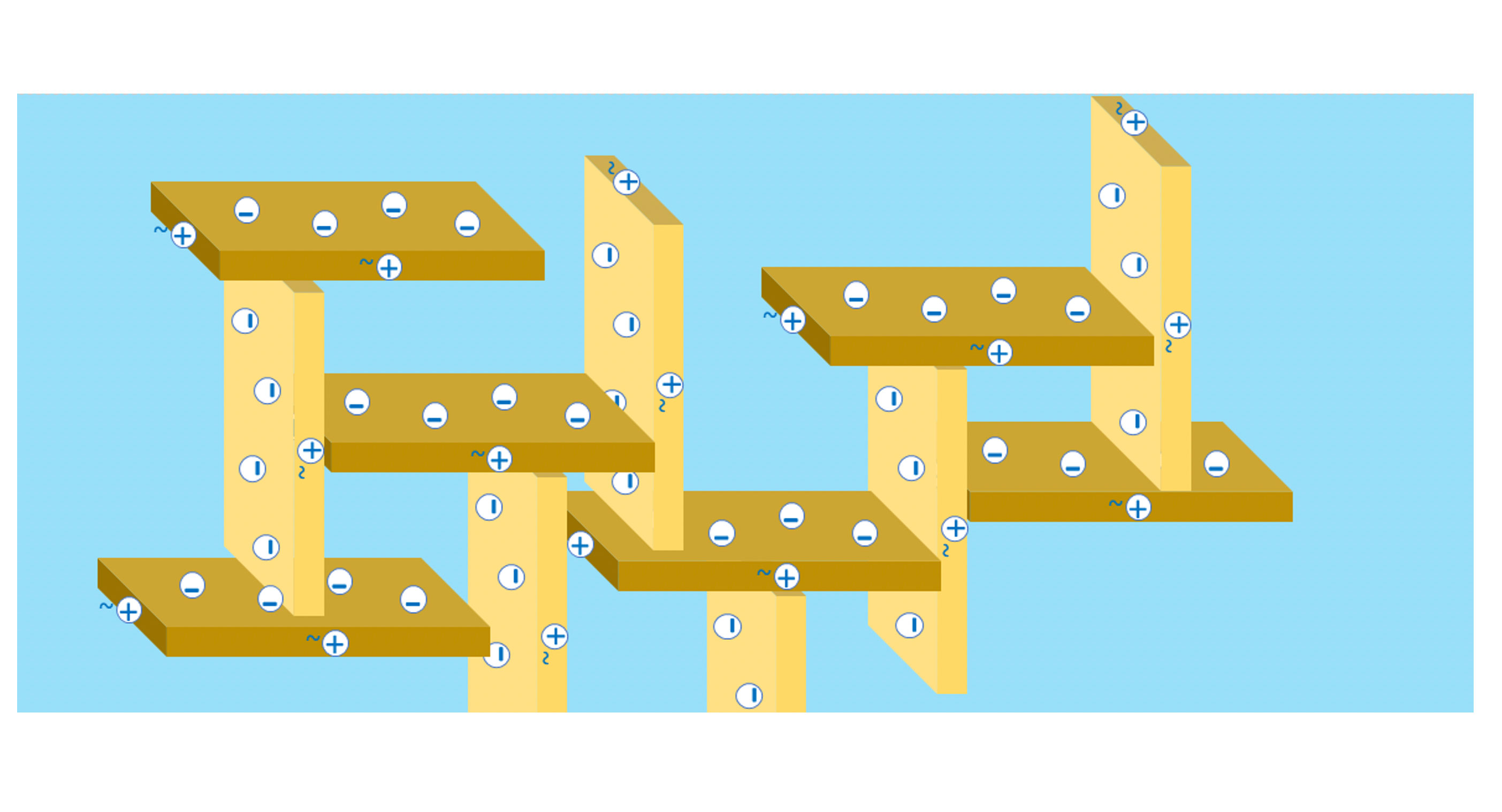
As the shear forces subside, positively charged surfaces become attractive to negatively charged surfaces. As a result, a “house of cards” structure is formed and a gel is formed [Figure 4]. The formed structure creates traps in which gases (e.g. air), solid particles or emulsions can be present. As a result, there may be an additional viscosity-increasing effect between the raw materials present in the paint formulation by trapping particles of other raw materials in the structure of the “house of cards”.
The resulting structure is easy to break using shear forces [Figure 5]. During mixing (shear thinning), the plates loosen and the viscosity drops, which is reversible. Thanks to this, mineral thickeners are pseudoplastic, and some of them are even thixotropic (the return to the original structure takes place with a delay in time).
So, if the basis for the hydration of mineral thickeners is the replacement of ions with water particles, does the purity of the water matter for the proper hydration? Can the presence of ions in the water increase or decrease the degree of hydration? I will answer these questions in this article.
EXPERIMENTAL
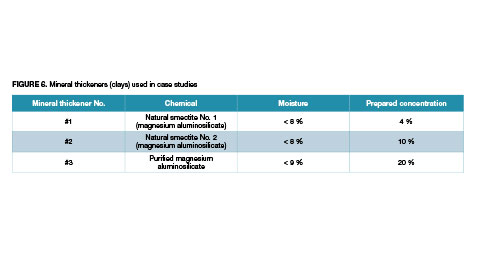
Case studies were carried out to determine the effect of water on hydration and its subsequent effect on the properties of latex paints prepared on a laboratory scale. Three mineral thickeners from different manufacturers from the US market were used for the tests
The assumption of the project was to check:
• the importance of the type of water for the hydration efficiency of mineral thickeners, and
• the impact of the type of water on the results of laboratory tests of paints with individual thickeners
The humidity of each sample was determined with the use of a moisture analyzer and assumed for the preparation of the concentration of dry thickener weights. The different concentrations in the water were selected on the basis of preliminary tests and the possibility of preparing a specific concentration.
Each of the samples was prepared in the cowles laboratory dissolver by dispersing in water:
• DI water type II according to ASTM D1193 (conductivity < 1 µS/cm)
• Tap water, medium hard 30 °f (ca. 17.5 °a), 3.0 mmol Ca2+
Samples of the clay suspensions were dispersed in the cowles dissolver at a relatively low peripheral speed (2.8 to 3.9 m/s), which was desirable to observe a more pronounced effect of water quality on the degree of hydration.
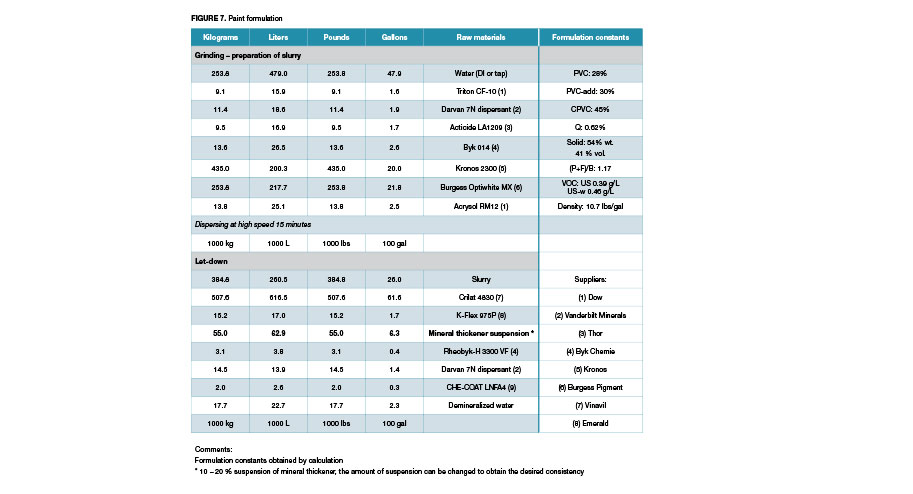
The prepared suspensions were used for the preparation of latex paints from the formulation as in Figure 7. The paint preparation consistently used the same type of water as used in the
clay suspension.
TEST RESULT
Below are the results of tests carried out within the scope of the project.
KU VISCOSITY
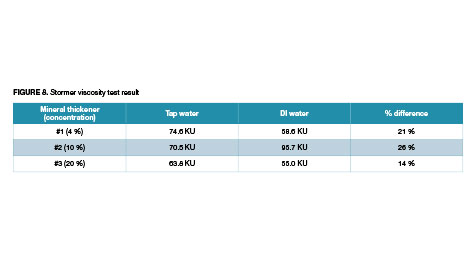
The determination was made for clays suspensions and for paints. The results are shown in Figure 8.
Figure 9 shows a photo of mineral thickener #1. On the left, a suspension in tap water, on the right, in demineralized water. There is a clear difference in sample liquidity.
BROOKFIELD VISCOSITY
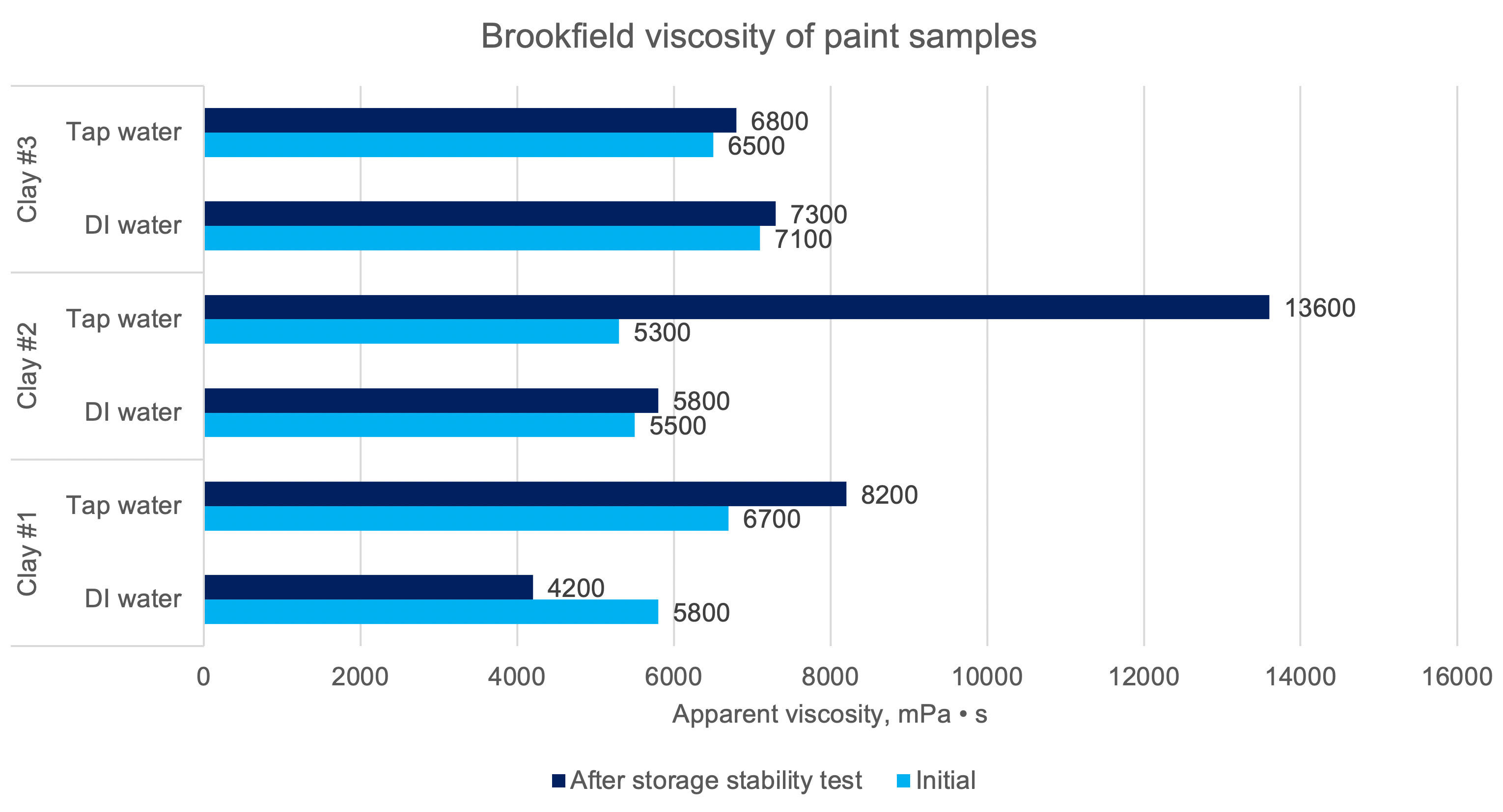
Apparent viscosity determinations were carried out for the inks subjected to the storage stability test (1 mount at 52 °C / 125 °F) with respect to the viscosity of the inks 72 hours after their preparation. The results are shown in Figure 10.
The paints have more or less the same initial viscosity. Their change, of course, occurs as a result of reactions also taking place between other components, however, the variable between the samples was demineralized water and the thickener and minerals, which in this case have the greatest impact on the observed changes. Clay #1 has a viscosity decrease of 27 % with demineralized water, while the viscosity increases after storage stability test for a sample with tap water by 22 %. Clay #2 paint shows very little viscosity difference for DI water, only a 5 % increase, which cannot be said for the clay #2 sample and tap water which shows a 156 % increase in viscosity. The clay #3 paint, on the other hand, showed almost no change in viscosity after the storage stability test for both DI and tap water.
SAGGING
The sagging resistance test results were performed by determining the Anti-Sag Index according to ASTM D4400 using a notched applicator with gaps ranging from 4 to 24 mils. The results are shown in Figure 11 with the Anti-Sag Index results.
In the case of sagging, differences can be seen for mineral thickener clay #1, where there is a difference of 2.8 mils between DI water and tap water. A sample with DI water has almost 16 % better sagging resistance than paint with tap water. in the case of clay #2, a slightly better result was obtained for the sample with tap water, as in the case of clay #3.
SUMMARY
The presented case study of three mineral thickeners in two types of water showed that there is an influence of water purity on the course of hydration, which in turn has an impact on some properties of paints. Of course, as I mentioned before, the effect of DI water and tap water on the remaining raw materials in the formulation is not indifferent, however, the variable water and clays showed a clear effect that is closely related to the type of thickener and there is no rule as to whether DI water or faucet is a better option. This is why the example of a paint producer mentioned in the introduction, who used the same raw materials, the same formulation but with water of different hardness, obtained different results of the viscosity and rheological behavior of paints in two factories.
The ions present in the water, as well as their lack or strong limitation, as is the case with demineralized water, affect many raw materials and their effectiveness in the formulation, e.g. dispersants or defoamers. The above-mentioned example concerning mineral thickeners proved that also. So if you’re formulating with mineral thickeners, check to see if your sample works more effectively in DI water, whether it’s hard, soft or very hard. For optimal efficiency and sustainable development.
Mineral thickeners are a group of organoclays that occur naturally in the form of deposits. They are made of laminar plates and are phyllosilicates used in a wide range of industries, from sorbents to cat litter. Due to their high ability to absorb liquids and form gels, they are also used as paint thickeners. Naturally occurring and modified bentonites, attapulgites, smectites and layered aluminosilicates are of particular importance.
Mineral thickeners are a very important group of rheological modifiers of architectural paints. They are responsible for the rheological properties of latex paints in the low (Brookfield-viscosity) and mid-shear (Stormer-viscosity) areas and for the synergy of action with rheology modifying thickeners in the areas of high shear forces (ICI-viscosity). In the area of low and mid-shear rates, they are responsible for imparting viscosity and maintaining the rheological properties of sagging, settling and sedimentation, storage stability, including syneresis prevention and the “feeling” of in-can viscosity. They work very well with other thickeners responsible for rheology in the area of low and medium shear rates, incl. cellulose derivatives and high-shear ICI-builders in elimination
of spattering.
Their ability to absorb liquids and form gels is closely related to their structure and the possibility of changes that occur during their swelling in the water environment. But is this swelling the same in all water? After all, as you know, water-based paints are made of both demineralized and tap water. Different producers also use post-process water (waste-water, recycled water), and the tap water has a different hardness, which in turn is related to different ions in its composition. This article discusses the basic influence of the type of water on the hydration of mineral thickeners, as well as their influence on the properties of paints prepared from them. I was inspired to write this article by the story of one of the paint manufacturers who, after opening a latex paint factory in another part of the country and despite using the same raw materials, could not obtain the same viscosity from the same formulation as in his other factory.
HYDRATION AND HOUSE OF CARDS
Mineral thickeners, e.g. smectites, are made of layers of platelets with dimensions of approximately 100 x 100 nm and a thickness of approximately 1 nm (0.001 µm). Their edges are slightly positively charged, while their platelet faces are negatively charged [Figure 1]. The negative surface charge is balanced by sodium ions, which are exchangeable and can be substituted by other cations, e.g. NH+, Ca2+ [Figure 2]. A single grain of clay is made up of thousands of sandwich platelet with exchangeable ions and a monomolecular water layer in between each. When the clay is dispersed in the water, the water penetrates the spaces between the platelets. Then ions are exchanged and replaced with water molecules (diffusion and osmosis), which results in delamination of the plates [Figure 3].
As the shear forces subside, positively charged surfaces become attractive to negatively charged surfaces. As a result, a “house of cards” structure is formed and a gel is formed [Figure 4]. The formed structure creates traps in which gases (e.g. air), solid particles or emulsions can be present. As a result, there may be an additional viscosity-increasing effect between the raw materials present in the paint formulation by trapping particles of other raw materials in the structure of the “house of cards”.
The resulting structure is easy to break using shear forces [Figure 5]. During mixing (shear thinning), the plates loosen and the viscosity drops, which is reversible. Thanks to this, mineral thickeners are pseudoplastic, and some of them are even thixotropic (the return to the original structure takes place with a delay in time).
So, if the basis for the hydration of mineral thickeners is the replacement of ions with water particles, does the purity of the water matter for the proper hydration? Can the presence of ions in the water increase or decrease the degree of hydration? I will answer these questions in this article.
EXPERIMENTAL
Case studies were carried out to determine the effect of water on hydration and its subsequent effect on the properties of latex paints prepared on a laboratory scale. Three mineral thickeners from different manufacturers from the US market were used for the tests
The assumption of the project was to check:
• the importance of the type of water for the hydration efficiency of mineral thickeners, and
• the impact of the type of water on the results of laboratory tests of paints with individual thickeners
The humidity of each sample was determined with the use of a moisture analyzer and assumed for the preparation of the concentration of dry thickener weights. The different concentrations in the water were selected on the basis of preliminary tests and the possibility of preparing a specific concentration.
Each of the samples was prepared in the cowles laboratory dissolver by dispersing in water:
• DI water type II according to ASTM D1193 (conductivity < 1 µS/cm)
• Tap water, medium hard 30 °f (ca. 17.5 °a), 3.0 mmol Ca2+
Samples of the clay suspensions were dispersed in the cowles dissolver at a relatively low peripheral speed (2.8 to 3.9 m/s), which was desirable to observe a more pronounced effect of water quality on the degree of hydration.
The prepared suspensions were used for the preparation of latex paints from the formulation as in Figure 7. The paint preparation consistently used the same type of water as used in the
clay suspension.
TEST RESULT
Below are the results of tests carried out within the scope of the project.
KU VISCOSITY
The determination was made for clays suspensions and for paints. The results are shown in Figure 8.
Figure 9 shows a photo of mineral thickener #1. On the left, a suspension in tap water, on the right, in demineralized water. There is a clear difference in sample liquidity.
BROOKFIELD VISCOSITY
Apparent viscosity determinations were carried out for the inks subjected to the storage stability test (1 mount at 52 °C / 125 °F) with respect to the viscosity of the inks 72 hours after their preparation. The results are shown in Figure 10.
The paints have more or less the same initial viscosity. Their change, of course, occurs as a result of reactions also taking place between other components, however, the variable between the samples was demineralized water and the thickener and minerals, which in this case have the greatest impact on the observed changes. Clay #1 has a viscosity decrease of 27 % with demineralized water, while the viscosity increases after storage stability test for a sample with tap water by 22 %. Clay #2 paint shows very little viscosity difference for DI water, only a 5 % increase, which cannot be said for the clay #2 sample and tap water which shows a 156 % increase in viscosity. The clay #3 paint, on the other hand, showed almost no change in viscosity after the storage stability test for both DI and tap water.
SAGGING
The sagging resistance test results were performed by determining the Anti-Sag Index according to ASTM D4400 using a notched applicator with gaps ranging from 4 to 24 mils. The results are shown in Figure 11 with the Anti-Sag Index results.
In the case of sagging, differences can be seen for mineral thickener clay #1, where there is a difference of 2.8 mils between DI water and tap water. A sample with DI water has almost 16 % better sagging resistance than paint with tap water. in the case of clay #2, a slightly better result was obtained for the sample with tap water, as in the case of clay #3.
SUMMARY
The presented case study of three mineral thickeners in two types of water showed that there is an influence of water purity on the course of hydration, which in turn has an impact on some properties of paints. Of course, as I mentioned before, the effect of DI water and tap water on the remaining raw materials in the formulation is not indifferent, however, the variable water and clays showed a clear effect that is closely related to the type of thickener and there is no rule as to whether DI water or faucet is a better option. This is why the example of a paint producer mentioned in the introduction, who used the same raw materials, the same formulation but with water of different hardness, obtained different results of the viscosity and rheological behavior of paints in two factories.
The ions present in the water, as well as their lack or strong limitation, as is the case with demineralized water, affect many raw materials and their effectiveness in the formulation, e.g. dispersants or defoamers. The above-mentioned example concerning mineral thickeners proved that also. So if you’re formulating with mineral thickeners, check to see if your sample works more effectively in DI water, whether it’s hard, soft or very hard. For optimal efficiency and sustainable development.