Gregory Monaghan, Specialty Polymers, Inc.07.05.17
Abstract
Concrete is a widely used and versatile construction material but, for some applications, using polymeric coatings or additives can significantly improve durability, adhesion or appearance. Improper substrate preparation or mistakes in the selection of coatings can lead to widespread (and expensive) coating failure. A background on cement chemistry, uses for polymeric additives and coatings, and the potential failure modes of coatings on concrete are presented in this paper. Several different coatings for concrete are discussed along with key advantages and disadvantages of the different chemistries.
Introduction
Concrete is the most widely used building material because it has many unique properties which make it ideal for use in construction jobs. It can be formed into different shapes on the job site, is very durable and requires little maintenance to withstand UV, mold or insects. Concrete is noncombustible and is relatively inexpensive relative to other building materials. It is also a particularly strong building material with high compressive strength.
The compressive strength of concrete is built slowly over a period of time, and can continue to increase as long as there is unreacted cement and water present. Three factors are necessary in developing the maximum strength – the correct combination of ingredients, the correct method of placement of the concrete and the correct curing. If any of these factors is not done correctly, then the strength of the concrete can be as much as 50% lower than expected. In addition, concrete can lose strength through chemical attack. Many of these failures can be eliminated by changing the concrete formulation to reduce the porosity of the cured concrete – or by using polymeric coatings or admixes which can give a better cure and/or protect from chemical attack.
Concrete Composition
Concrete is composed of cement, water and aggregate. Aggregate is the sand and gravel used in concrete – composing 60 to 75% of the concrete. Like the extender pigments used in paints, the aggregate is a relatively low-cost ingredient (relative to the cement) and it keeps the cost of the concrete low. Both fine aggregate (typically sand, but can be crushed rock with a particle size of less than 3/8 inch) and coarse aggregate (typically gravel with a particle size of less than 1.5 inches) are used. The aggregate plays an important role in preventing shrinkage of the paste surrounding the aggregate during drying which can cause cracking. Similar to the concept of PVC in paint, the larger the aggregate size, and the more rounded the aggregate, the lower the amount of cement and water required since more cement paste is required to coat the surface area of smaller or more angular aggregate.
Cement is a man-made material and the highest cost component of concrete but since it is usually only about 15% of the mix, the overall cost of the concrete is low. Cement is formed during a high-temperature calcining process during which minerals like clay, iron ore, sand and limestone are partially melted and recombined at up to 2,700°F in large kilns wide enough to fit a car and as long as a 40 story building. During a preheating step at 900°F, the calcium carbonate is converted to calcium oxide (lime) and carbon dioxide is driven off. As the temperature is slowly increased to 1500°F, the aluminate and ferrite phases melt and some of the calcium oxide reacts with the silica oxide to form a calcium silicate mineral called belite. As the temperature is increased further to 2700°F, much of the belite is converted to a more reactive form of calcium silicate called alite which is key to the early strength of portland cement. The silicates are cooled rapidly to form gray softball-size lumps called “clinker” which is ground to form sub-micron size particles. The high-temperature calcining process results in cement particles which are soluble and unstable at room temperature when they become wet. In the hydration process, the cement particles form a paste which surrounds the aggregates. This process involves the dissolving of the cement particles in the water, followed by the precipitation of the hydrates (which form the paste) from a super saturated solution.
The precipitated hydrates are not the same materials which first dissolved. There is water associated with the precipitates which forms hydrates. Some of these hydrates are larger in volume than the dry cement particles and they continue to form and occupy space that was occupied by the free water in the concrete. In addition, the precipitates are more energetically favored than the cement and, as a result, there is a release of heat – the heat of hydration – which can accelerate the hydration reaction.
Although cement is relatively expensive, the total amount of cement in concrete can be reduced by the addition of some recycled and low cost ingredients like fly ash formed in the coal fired power plants. Fly ash has a pozzolanic activity (when finely divided it reacts with water and calcium from lime to form a compound with cementitious properties) and can reduce the amount of expensive cement in the mixture by up to 30%. Fly ash is formed during the burning of coal at temperatures of 2,700°F – it is a finely divided alumino silicate glass that is carried off by flue gasses and collected electrostatically. Because the particles are melted and reformed in the gas phase, they are spherical. The shape and composition have a lot to do with the beneficial properties fly ash brings to the concrete. Because of the spherical shape, fly ash can reduce the amount of water required for good workability which contributes to higher strength when cured.2 In large pours of concrete where heat dissipation is a potential problem (temperature differentials can lead to concrete cracking), the fly ash can significantly reduce the heat generated. Most significantly, however, fly ash also replaces part of the expensive cement (typically up to 30%) which reduces the cost of the concrete, while also giving improved performance. There is one significant drawback, however. The solubility of the fly ash is less than some of the other components of concrete and it reacts more slowly. As a result, fly ash will significantly slow the cure time of the concrete, which means that contractors have to wait longer to finish a job. Despite this downside, the overall benefits of fly ash are significant enough that it is in common use in concrete.
The amount of water is also key to the properties of concrete. The strength of the concrete depends to a large extent on the amount of water relative to the cement (w/c ratio).
This is a balance that has an optimum w/c ratio, however. Free water must be present for the hydrates to form and the concrete to gain maximum strength. Although the water is essential to the formation of the hydrates, too much water can detract from the strength because it can create unfilled voids in the concrete. There is a set amount of water which is required to form the calcium silicate hydrates, but extra water is needed to make the concrete workable. Too little water will give a mixture which is difficult to pour out and work; if it is difficult to compact, then the strength will also be lower because of air voids. A quality concrete is made by using as little water as possible but still maintaining enough water for good workability. If too much water is added at the job site to make the concrete easier to work when it is being poured, the concrete will have voids in it which are not filled by the hydrates, and the strength will be lower.
Cement Components
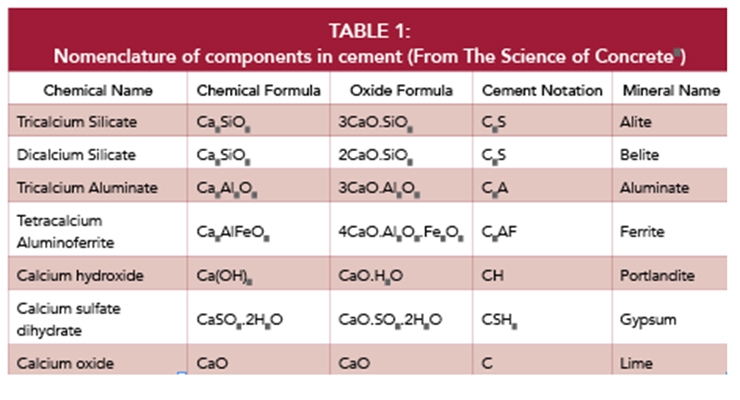
To understand the formation of hydrates which are the key to the strength of concrete, we need to look at the different components of cement individually, although the actual reactions result in a mixed product of each of the following chemicals. Cement chemists use a different notation to describe the materials.
Alite
The alite is the most abundant mineral in portland cement at 40-60% of the total. It is the hydration reaction of the alite with water which gives concrete its initial strength. Alite can form several crystal structures, however when it is cooled rapidly after calcining, it forms an awkward structure in which the calcium and oxygen do not fit together well in the crystal matrix. This gives alite its high reactivity and solubility. The alite rapidly dissolves in the water phase and, after the solution becomes super saturated, it precipitates out as a calcium silicate hydrate gel (C-S-H).1
The C-S-H hydrate gel which forms is not intrinsically strong, but it forms a layer around other particles in the concrete and this binds the individual particles together. As the CSH gel forms it contains millions of tiny water filled voids in the gel matrix (much smaller than the water filled pores in the concrete). This CSH gel with the micro voids has a much larger volume than the original tricalcium oxide silicate particles. As a result the CSH gel expands outward and encapsulates the other aggregate particles in the concrete. This causes the concrete to set and eventually to harden to a strong solid. During this process the water filled capillary pores in the concrete get smaller or close up. This reduction in the volume of the pores decreases the permeability of the concrete.
The calcium hydroxide (Portlandite) which forms during this hydration reaction contributes only slightly to the strength of the concrete. It reacts with liquid water and helps close the pores and it forms individual crystals which are resistant to shrinkage during drying. The CH is water soluble, however, and if the concrete is exposed to fresh water, the CH will leach out, leading to larger pores.
Belite
Belite is the name of the dicalcium oxide silicate which is also present in cement after the calcination. It is formed at lower temperatures than the alite and early concrete (created in lower temperature kilns) was almost exclusively belite. Belite is much less soluble than alite and doesn’t react as fast. Because of the slower hydration rate, belite contributes little to the initial strength of concrete but it does build over time and it is a major contributor to the mature strength of concrete.
Aluminate and Ferrite
Portland cement also contains tricalcium aluminate and tetracalcium alumina ferrite. These are essential to the formation of the liquid phase in the calcining process since they melt at lower temperatures; the liquid from the melted aluminates hold the solid belite particles as they are slowly converted to the more reactive alite. The calcium aluminate is as soluble as the C3S and it reacts rapidly to form a mineral called hydrogarnet.
This reaction releases so much heat that if it were allowed to take place the concrete would set in minutes, too fast for good workability. To prevent this, a small amount of gypsum is added to cement when the clinker is ground. The gypsum is also highly soluble and the sulfates react with the aluminates to form the mineral ettringite. If all the sulfate reacts before all the C3A does then the level of the sulfate ions decreases and the ettringite becomes unstable and undergoes a second reaction over several hours to form monosulfate which is lower in sulfate. Because there is not enough gypsum to fully react with the C3A, there is rarely ettringite in cured concrete.
The ferrite phase (C4AF) in the cement has some of the aluminum substituted with iron. It is less soluble than the aluminate and, as a result, it reacts more slowly. Both the aluminate and the ferrite phase contribute to the heat of hydration and affect the rheology of the setting concrete. In addition, the products of the hydration of the ferrite phase can contribute to the compressive strength of the concrete.7
Induction Period
There is a short period of several hours after the addition of water during which the rate of hydration nearly stops; this is called the induction period. The fact that this induction period exists is critical to the successful use of concrete. It means that the concrete truck can be filled and sent to a job site and there still be enough time to apply the wet concrete before it sets. Concrete chemists disagree on the cause of the induction period, however the most common theory is that an unstable gel or (in the case of the aluminate phase) ettringite is formed around the cement particles C3S and C3A. This unstable gel prevents the cement particles from further dissolving in the pore solution slowing the reaction in the first few hours after mixing. With time these unstable gels are converted to other species which allow the cement particle to continue to dissolve, speeding the reaction again.
Hydration Reactions
Hydration reactions will continue as long as there is free water, space in the pores for the hydrates to grow, and unhydrated silicates present (the core of the undissolved cement particles). Although most of the hydration takes place in the first month, the hydration reactions continue for months or years. Different components of concrete contribute to cure at different times, however. After 6-8 hours the ettringite, calcium hydroxide and C-S-H gel are being formed and the porosity begins to drop as the gel forms in the water filled pores. The slower reacting ferrite species only begins to react after several days. After a couple days, the concentration of the ettringite levels off then falls as the gypsum is consumed and monosulfate is produced.8
If the concrete dries out prematurely, the hydration reactions will stop and the concrete cannot build any more strength. In addition, early drying can lead to stress cracking since the concrete will shrink slightly as it dries, which can create stresses at the surface (since the interior of the concrete doesn’t dry at the same rate as the outside). If the concrete can be kept wet, the strength will build enough that the concrete can resist the cracking. As a result, it is common to try to restrict the loss of humidity and heat from the concrete in the critical early stages of cure – usually the first 6 days after the pour. There are several ways of doing this.
One method of keeping the concrete surface moist is to mist the surface with water or pond the water on the surface. This is low cost, although it requires a significant amount of water to mist the surface for several days. Misting the surface can also be difficult on vertical concrete structures and coatings are often used instead to slow water loss on those structures.
The most common method of keeping the surface moist is to use polyethylene sheeting over wet burlap, however this method can lead to several problems. There is also the chance of staining the concrete if the sheeting is not evenly applied and, if the burlap is not kept wet, it can wick water from the concrete, leading to cracking. Unless the sheeting is well attached, wind can get underneath and lift the sheeting creating a wind tunnel and drying the surface even more. As a result, this method is somewhat labor intensive with the possibility of damage to the concrete.
Coatings for Concrete
There are also several coatings which can be used to help slow the loss of moisture and help cure the concrete. These can be temporary or permanent coatings.
Temporary Coatings
Temporary coatings are designed to help keep the moisture in the concrete for the first week or so but then to degrade under UV exposure, abrasion from traffic, or removal by using special cleaners. They have traditionally been coal tar resins, but are now more commonly hydrocarbon resins and supplied either in solvent or emulsified in water. The coatings are usually spray applied once the bleed water has evaporated but before the concrete has had a chance to dry. These coatings need to be removed if a coating is to be applied to the concrete after cure because they can interfere with the adhesion of the topcoat. Since they often do not degrade evenly, mechanical or chemical removal is often used.
Wax emulsion coatings (typically paraffin emulsions or lower-cost slack wax emulsions with some resin) are also temporary coatings used to prevent water loss. While effective at reducing moisture loss, they can cause discoloration on aging and they also have to be removed if a coating is to be applied to the concrete later. If they are not removed they can interfere with the adhesion of topcoats which are applied later.
Permanent Coatings
There are also more permanent coatings called cure and seal coatings. These are typically water-based styrene acrylic or acrylic emulsions which are applied while the concrete is wet but then form a more permanent film over the concrete. These coatings slow moisture loss during the early stages of curing, but they also have some benefits as sealers.
They can give protection from stains and can help minimize dusting from floor abrasion. In addition, they can give protection from some types of degradation caused by moisture and certain salts. Cure and seal coatings are tested by measuring the weight loss of water from a coated, curing concrete test block. ASTM C309 specifies less than 0.55 kg/m2 of water loss, and the more restrictive ASTM C1315 specifies less than 0.40 kg/m2 over 72 hours (in addition to UV durability and non-yellowing characteristics). Specially designed styrene acrylics can meet the ASTM C309 specifications at a relatively low cost and acrylics are often used in the more durable, non-yellowing cure and seal coatings designed to meet ASTM C1315 Class A.
Failure Modes
Although it is very durable under UV light and plain water, cured concrete is subject to several types of chemical attack which can degrade the surface or cause cracking. In most cases salts carried into the concrete by water can cause the degradation.
When there is an outside source of sulfate ions from ground water or exposure to sea water, then ettringite can be reformed from the monosulfate. This is an expansive process because the ettringite is a larger volume than the monosulfate. The expansive sulfate attack reaction puts stresses on the concrete and can lead to cracking, a loss of the bond between the cement paste and the aggregate and this results in a significant loss of strength. Resistance to sulfate attack is built through the use of low water/cement ratios (lower porosity), the use of silica fume or fly ash possolans, or by using surface coatings.8
Concrete is also subject to degradation from an alkali silicate reaction (ASR) if there is incorrect selection of aggregate. Some silicate minerals used as aggregate will react with sodium or potassium alkali in high pH conditions to form a higher-volume product which can put expansive stresses on the concrete and cause cracking. While the alkali metals sodium and potassium that are present in cement are typically low, external sources of alkali like deicing salts or sea water can lead to ASR. Reducing the ingress of external alkalis through less permeable concrete (lower w/c) or using coatings or sealers can reduce the potential for the ASR.3
Chloride ions can also be a significant problem in steel reinforced concrete. Steel reinforcement rods in wet concrete quickly form a protective rust layer at the surface which protect the rods from further degradation. The chloride ions destroy the protective Fe2O3 passivating layer on steel and, if there is water and air present in the concrete pores, then an electrochemical current is set up and rust is formed at the anode. In addition to causing pitting in the steel, the rust has a larger volume than the steel and it gives expansive pressure on the surrounding concrete and can cause cracking of the concrete.5 Decreasing the porosity with a lower water to cement ratio and the use of fly ash can mitigate chloride attack, however these preventative measures are often used in conjunction with concrete coatings or coatings for the steel surface.
If deicing salts are used, they can collect in the voids at the surface of the concrete. The crystallization of the salts can put stress on the surface layers of concrete and cause scaling. This is worse in concrete exposed to repeated wet/dry cycles. Coatings which protect the surface from moisture can prevent this type of degradation.4
Concrete can also degrade on exposure to acid. Acids can dissolve both hydrated and unhydrated cement compounds and can dissolve calcium carbonate (calcareous) based aggregate. Acid rain (pH 4.5-5) can lead to slight etching of the surface but does not significantly affect performance. Reducing the porosity of the concrete with coatings can reduce the degradation.4
Another common concrete failure is caused by freeze thaw cycles. The freezing of water in the pores creates stresses in the concrete which can lead to spalling. It is common to use air entraining additives when mixing wet concrete to prevent damage from freeze thaw cycles. The entrained air pockets in the dried concrete give water room to expand and prevent damage from freezing. Another way to help prevent damage is to reduce the porosity which can help prevent the ingress of water. The porosity can be reduced by reducing the water to cement ratio as much as possible or by using sealers.
Sealers
Polymeric sealers can help prevent the chemical or physical degradation of concrete. Sealers are typically described as penetrating or film forming and are differentiated from cure and seal coatings since they are applied after the concrete has cured for 28 days.
Silanes or siloxanes are penetrating sealers. They do help seal the surface against liquid water and stains but they do not prevent water vapor loss from the concrete during cure. The silanes have a low molecular weight while the siloxanes are prereacted and have an EO chain giving higher molecular weight. Because of the lower molecular weight, the silanes can give more penetration into the pores than the silanes. Since they are beneath the surface, they are more abrasion resistant than the aqueous sealers which form a film on the surface.
Aqueous emulsions used as sealers can also help prevent ingress of salts and moisture which can lead to degradation. These are typically acrylic or styrene acrylic and have good resistance to liquid water but are breathable and allow vapor to pass through the film. In addition to water resistance, water whitening resistance is also needed to avoid discoloration if exposed to puddled water. Polymers need to be designed to be blush resistant by keeping the particle size small, using special polymerizable surfactants and keeping the level of other hydrophilic materials (such as surfactants) low. These emulsion sealers are typically designed to be applied at low dry film thicknesses and they are typically not high gloss finishes – usually satin or semigloss finishes. Since the films are relatively thin and largely on the surface of the concrete, they usually need to be reapplied every other year to maintain protection.
Solvent-based acrylics are also commonly used as sealers and are usually applied after the concrete has aged for a month. These sealers are very durable because of the acrylic backbone, but still low enough molecular weight to penetrate into the concrete pores and provide a darkening of the surface (called a wet look) which can be desirable. They are applied at higher film thicknesses than the acrylic emulsions and can give a high gloss when dried. These acrylics can be formulated in exempt solvents like acetone, however the rapid dry speed and odor can present a significant issue during application.
Two part epoxies are used to seal concrete in environments where a high level of chemical resistance is needed. These can be cured with polyamines, polyamides or polysulfides, although the polyamide-cured epoxies typically have lower chemical resistance. The epoxies have good adhesion to the concrete and can be formulated to be relatively oil tolerant for use on garage floors, or designed to cure at low temperatures. Epoxies can chalk on exterior exposure, however, and they are not usually used for outdoor applications.
In the case of chloride attack on the reinforcing steel, it is also possible to reduce the effect by applying a thin overlayment of polymer modified cement to reduce the surface porosity of concrete. The polymeric admixes function by reducing the amount of water necessary for a given concrete fluidity resulting in a lower water to cement ratio. In addition, as the concrete dries the polymer forms films in the pores and blocks the capillaries. The slower cure of the concrete and the dried polymer in the matrix results in reduced porosity.6 While the reduced porosity of the overlayment helps reduce the penetration of water carrying chloride ions, the polymer can also improve adhesion to the concrete substrate and can improve durability by increasing the tensile strength, resistance to abrasion and dusting.
Summary
In order for the complex chemical reactions in concrete to occur, water is required. To build optimum strength and avoid cracking at the surface, it is also necessary to keep concrete moist for several days after being poured. Polymeric coatings and cure and seal coatings can help develop the strength of concrete by reducing the porosity at the surface and keeping the water in the concrete longer. Cured concrete is also subject to chemical attack or physical degradation by freeze thaw cycles. Sealers or polymer-modified overlayments applied after cure can help protect concrete from these forms of degradation.
References
1. Thomas, J.; Jennings, H. (n.d.). Science of Concrete. Retrieved June 28, 2016, from http://iti.northwestern.edu/cement/index.html
2. Thomas, M. (2007). Optimizing the use of Fly Ash in Concrete. Retrieved July 15, 2016, from http://www.cement.org/docs/default-source/fc_concrete_technology/is548-optimizing-the-use-of-fly-ash-concrete.pdf
3. Farny, J.; Kerkhoff, B. (2007). Diagnosis and Control of Alkali-Aggregate Reactions in Concrete. Retrieved June 14, 2016, from http://www.cement.org/docs/default-source/fc_concrete_technology/is413-02---diagnosis-and-control-of-alkali-aggregate-reactions-in-concrete.pdf?sfvrsn=2
4. Kerkhoff, B. (2007). Effects of Substances on Concrete and Guide to Protective Treatments [Pamphlet]. Skokie, IL: Portland Cement Association.
5. Neville, A. (1995). Chloride attack of reinforced concrete: An overview. Materials and Structures, 28, 63-70.
6. ATS 11 - Polymer Dispersion Admixtures. (2016, April 21). Retrieved June 14, 2016, from http://www.admixtures.org.uk/download/ats-11-polymer-dispersion-admixtures/
7. Duvallet, T. (2014). Influence Of Ferrite Phase In Alite Calcium Sulfoaluminate Cements (Published doctoral dissertation, 2014). University of Kentucky. Retrieved August 8, 2016, from http://uknowledge.uky.edu/cme_etds/27/
8. Kurtis, K. (2015) Portland Cement Hydration. Retrieved July 7, 2016 from http://people.ce.gatech.edu/~kk92/hyd07.pdf
9. Stark, D. (2002) Performance of Concrete in Sulfate Environments, [Pamphlet]. Skokie, IL: Portland Cement Association.
Concrete is a widely used and versatile construction material but, for some applications, using polymeric coatings or additives can significantly improve durability, adhesion or appearance. Improper substrate preparation or mistakes in the selection of coatings can lead to widespread (and expensive) coating failure. A background on cement chemistry, uses for polymeric additives and coatings, and the potential failure modes of coatings on concrete are presented in this paper. Several different coatings for concrete are discussed along with key advantages and disadvantages of the different chemistries.
Introduction
Concrete is the most widely used building material because it has many unique properties which make it ideal for use in construction jobs. It can be formed into different shapes on the job site, is very durable and requires little maintenance to withstand UV, mold or insects. Concrete is noncombustible and is relatively inexpensive relative to other building materials. It is also a particularly strong building material with high compressive strength.
The compressive strength of concrete is built slowly over a period of time, and can continue to increase as long as there is unreacted cement and water present. Three factors are necessary in developing the maximum strength – the correct combination of ingredients, the correct method of placement of the concrete and the correct curing. If any of these factors is not done correctly, then the strength of the concrete can be as much as 50% lower than expected. In addition, concrete can lose strength through chemical attack. Many of these failures can be eliminated by changing the concrete formulation to reduce the porosity of the cured concrete – or by using polymeric coatings or admixes which can give a better cure and/or protect from chemical attack.
Concrete Composition
Concrete is composed of cement, water and aggregate. Aggregate is the sand and gravel used in concrete – composing 60 to 75% of the concrete. Like the extender pigments used in paints, the aggregate is a relatively low-cost ingredient (relative to the cement) and it keeps the cost of the concrete low. Both fine aggregate (typically sand, but can be crushed rock with a particle size of less than 3/8 inch) and coarse aggregate (typically gravel with a particle size of less than 1.5 inches) are used. The aggregate plays an important role in preventing shrinkage of the paste surrounding the aggregate during drying which can cause cracking. Similar to the concept of PVC in paint, the larger the aggregate size, and the more rounded the aggregate, the lower the amount of cement and water required since more cement paste is required to coat the surface area of smaller or more angular aggregate.
Cement is a man-made material and the highest cost component of concrete but since it is usually only about 15% of the mix, the overall cost of the concrete is low. Cement is formed during a high-temperature calcining process during which minerals like clay, iron ore, sand and limestone are partially melted and recombined at up to 2,700°F in large kilns wide enough to fit a car and as long as a 40 story building. During a preheating step at 900°F, the calcium carbonate is converted to calcium oxide (lime) and carbon dioxide is driven off. As the temperature is slowly increased to 1500°F, the aluminate and ferrite phases melt and some of the calcium oxide reacts with the silica oxide to form a calcium silicate mineral called belite. As the temperature is increased further to 2700°F, much of the belite is converted to a more reactive form of calcium silicate called alite which is key to the early strength of portland cement. The silicates are cooled rapidly to form gray softball-size lumps called “clinker” which is ground to form sub-micron size particles. The high-temperature calcining process results in cement particles which are soluble and unstable at room temperature when they become wet. In the hydration process, the cement particles form a paste which surrounds the aggregates. This process involves the dissolving of the cement particles in the water, followed by the precipitation of the hydrates (which form the paste) from a super saturated solution.
The precipitated hydrates are not the same materials which first dissolved. There is water associated with the precipitates which forms hydrates. Some of these hydrates are larger in volume than the dry cement particles and they continue to form and occupy space that was occupied by the free water in the concrete. In addition, the precipitates are more energetically favored than the cement and, as a result, there is a release of heat – the heat of hydration – which can accelerate the hydration reaction.
Although cement is relatively expensive, the total amount of cement in concrete can be reduced by the addition of some recycled and low cost ingredients like fly ash formed in the coal fired power plants. Fly ash has a pozzolanic activity (when finely divided it reacts with water and calcium from lime to form a compound with cementitious properties) and can reduce the amount of expensive cement in the mixture by up to 30%. Fly ash is formed during the burning of coal at temperatures of 2,700°F – it is a finely divided alumino silicate glass that is carried off by flue gasses and collected electrostatically. Because the particles are melted and reformed in the gas phase, they are spherical. The shape and composition have a lot to do with the beneficial properties fly ash brings to the concrete. Because of the spherical shape, fly ash can reduce the amount of water required for good workability which contributes to higher strength when cured.2 In large pours of concrete where heat dissipation is a potential problem (temperature differentials can lead to concrete cracking), the fly ash can significantly reduce the heat generated. Most significantly, however, fly ash also replaces part of the expensive cement (typically up to 30%) which reduces the cost of the concrete, while also giving improved performance. There is one significant drawback, however. The solubility of the fly ash is less than some of the other components of concrete and it reacts more slowly. As a result, fly ash will significantly slow the cure time of the concrete, which means that contractors have to wait longer to finish a job. Despite this downside, the overall benefits of fly ash are significant enough that it is in common use in concrete.
The amount of water is also key to the properties of concrete. The strength of the concrete depends to a large extent on the amount of water relative to the cement (w/c ratio).
This is a balance that has an optimum w/c ratio, however. Free water must be present for the hydrates to form and the concrete to gain maximum strength. Although the water is essential to the formation of the hydrates, too much water can detract from the strength because it can create unfilled voids in the concrete. There is a set amount of water which is required to form the calcium silicate hydrates, but extra water is needed to make the concrete workable. Too little water will give a mixture which is difficult to pour out and work; if it is difficult to compact, then the strength will also be lower because of air voids. A quality concrete is made by using as little water as possible but still maintaining enough water for good workability. If too much water is added at the job site to make the concrete easier to work when it is being poured, the concrete will have voids in it which are not filled by the hydrates, and the strength will be lower.
Cement Components
To understand the formation of hydrates which are the key to the strength of concrete, we need to look at the different components of cement individually, although the actual reactions result in a mixed product of each of the following chemicals. Cement chemists use a different notation to describe the materials.
Alite
The alite is the most abundant mineral in portland cement at 40-60% of the total. It is the hydration reaction of the alite with water which gives concrete its initial strength. Alite can form several crystal structures, however when it is cooled rapidly after calcining, it forms an awkward structure in which the calcium and oxygen do not fit together well in the crystal matrix. This gives alite its high reactivity and solubility. The alite rapidly dissolves in the water phase and, after the solution becomes super saturated, it precipitates out as a calcium silicate hydrate gel (C-S-H).1
The C-S-H hydrate gel which forms is not intrinsically strong, but it forms a layer around other particles in the concrete and this binds the individual particles together. As the CSH gel forms it contains millions of tiny water filled voids in the gel matrix (much smaller than the water filled pores in the concrete). This CSH gel with the micro voids has a much larger volume than the original tricalcium oxide silicate particles. As a result the CSH gel expands outward and encapsulates the other aggregate particles in the concrete. This causes the concrete to set and eventually to harden to a strong solid. During this process the water filled capillary pores in the concrete get smaller or close up. This reduction in the volume of the pores decreases the permeability of the concrete.
The calcium hydroxide (Portlandite) which forms during this hydration reaction contributes only slightly to the strength of the concrete. It reacts with liquid water and helps close the pores and it forms individual crystals which are resistant to shrinkage during drying. The CH is water soluble, however, and if the concrete is exposed to fresh water, the CH will leach out, leading to larger pores.
Belite
Belite is the name of the dicalcium oxide silicate which is also present in cement after the calcination. It is formed at lower temperatures than the alite and early concrete (created in lower temperature kilns) was almost exclusively belite. Belite is much less soluble than alite and doesn’t react as fast. Because of the slower hydration rate, belite contributes little to the initial strength of concrete but it does build over time and it is a major contributor to the mature strength of concrete.
Aluminate and Ferrite
Portland cement also contains tricalcium aluminate and tetracalcium alumina ferrite. These are essential to the formation of the liquid phase in the calcining process since they melt at lower temperatures; the liquid from the melted aluminates hold the solid belite particles as they are slowly converted to the more reactive alite. The calcium aluminate is as soluble as the C3S and it reacts rapidly to form a mineral called hydrogarnet.
This reaction releases so much heat that if it were allowed to take place the concrete would set in minutes, too fast for good workability. To prevent this, a small amount of gypsum is added to cement when the clinker is ground. The gypsum is also highly soluble and the sulfates react with the aluminates to form the mineral ettringite. If all the sulfate reacts before all the C3A does then the level of the sulfate ions decreases and the ettringite becomes unstable and undergoes a second reaction over several hours to form monosulfate which is lower in sulfate. Because there is not enough gypsum to fully react with the C3A, there is rarely ettringite in cured concrete.
The ferrite phase (C4AF) in the cement has some of the aluminum substituted with iron. It is less soluble than the aluminate and, as a result, it reacts more slowly. Both the aluminate and the ferrite phase contribute to the heat of hydration and affect the rheology of the setting concrete. In addition, the products of the hydration of the ferrite phase can contribute to the compressive strength of the concrete.7
Induction Period
There is a short period of several hours after the addition of water during which the rate of hydration nearly stops; this is called the induction period. The fact that this induction period exists is critical to the successful use of concrete. It means that the concrete truck can be filled and sent to a job site and there still be enough time to apply the wet concrete before it sets. Concrete chemists disagree on the cause of the induction period, however the most common theory is that an unstable gel or (in the case of the aluminate phase) ettringite is formed around the cement particles C3S and C3A. This unstable gel prevents the cement particles from further dissolving in the pore solution slowing the reaction in the first few hours after mixing. With time these unstable gels are converted to other species which allow the cement particle to continue to dissolve, speeding the reaction again.
Hydration Reactions
Hydration reactions will continue as long as there is free water, space in the pores for the hydrates to grow, and unhydrated silicates present (the core of the undissolved cement particles). Although most of the hydration takes place in the first month, the hydration reactions continue for months or years. Different components of concrete contribute to cure at different times, however. After 6-8 hours the ettringite, calcium hydroxide and C-S-H gel are being formed and the porosity begins to drop as the gel forms in the water filled pores. The slower reacting ferrite species only begins to react after several days. After a couple days, the concentration of the ettringite levels off then falls as the gypsum is consumed and monosulfate is produced.8
If the concrete dries out prematurely, the hydration reactions will stop and the concrete cannot build any more strength. In addition, early drying can lead to stress cracking since the concrete will shrink slightly as it dries, which can create stresses at the surface (since the interior of the concrete doesn’t dry at the same rate as the outside). If the concrete can be kept wet, the strength will build enough that the concrete can resist the cracking. As a result, it is common to try to restrict the loss of humidity and heat from the concrete in the critical early stages of cure – usually the first 6 days after the pour. There are several ways of doing this.
One method of keeping the concrete surface moist is to mist the surface with water or pond the water on the surface. This is low cost, although it requires a significant amount of water to mist the surface for several days. Misting the surface can also be difficult on vertical concrete structures and coatings are often used instead to slow water loss on those structures.
The most common method of keeping the surface moist is to use polyethylene sheeting over wet burlap, however this method can lead to several problems. There is also the chance of staining the concrete if the sheeting is not evenly applied and, if the burlap is not kept wet, it can wick water from the concrete, leading to cracking. Unless the sheeting is well attached, wind can get underneath and lift the sheeting creating a wind tunnel and drying the surface even more. As a result, this method is somewhat labor intensive with the possibility of damage to the concrete.
Coatings for Concrete
There are also several coatings which can be used to help slow the loss of moisture and help cure the concrete. These can be temporary or permanent coatings.
Temporary Coatings
Temporary coatings are designed to help keep the moisture in the concrete for the first week or so but then to degrade under UV exposure, abrasion from traffic, or removal by using special cleaners. They have traditionally been coal tar resins, but are now more commonly hydrocarbon resins and supplied either in solvent or emulsified in water. The coatings are usually spray applied once the bleed water has evaporated but before the concrete has had a chance to dry. These coatings need to be removed if a coating is to be applied to the concrete after cure because they can interfere with the adhesion of the topcoat. Since they often do not degrade evenly, mechanical or chemical removal is often used.
Wax emulsion coatings (typically paraffin emulsions or lower-cost slack wax emulsions with some resin) are also temporary coatings used to prevent water loss. While effective at reducing moisture loss, they can cause discoloration on aging and they also have to be removed if a coating is to be applied to the concrete later. If they are not removed they can interfere with the adhesion of topcoats which are applied later.
Permanent Coatings
There are also more permanent coatings called cure and seal coatings. These are typically water-based styrene acrylic or acrylic emulsions which are applied while the concrete is wet but then form a more permanent film over the concrete. These coatings slow moisture loss during the early stages of curing, but they also have some benefits as sealers.
They can give protection from stains and can help minimize dusting from floor abrasion. In addition, they can give protection from some types of degradation caused by moisture and certain salts. Cure and seal coatings are tested by measuring the weight loss of water from a coated, curing concrete test block. ASTM C309 specifies less than 0.55 kg/m2 of water loss, and the more restrictive ASTM C1315 specifies less than 0.40 kg/m2 over 72 hours (in addition to UV durability and non-yellowing characteristics). Specially designed styrene acrylics can meet the ASTM C309 specifications at a relatively low cost and acrylics are often used in the more durable, non-yellowing cure and seal coatings designed to meet ASTM C1315 Class A.
Failure Modes
Although it is very durable under UV light and plain water, cured concrete is subject to several types of chemical attack which can degrade the surface or cause cracking. In most cases salts carried into the concrete by water can cause the degradation.
When there is an outside source of sulfate ions from ground water or exposure to sea water, then ettringite can be reformed from the monosulfate. This is an expansive process because the ettringite is a larger volume than the monosulfate. The expansive sulfate attack reaction puts stresses on the concrete and can lead to cracking, a loss of the bond between the cement paste and the aggregate and this results in a significant loss of strength. Resistance to sulfate attack is built through the use of low water/cement ratios (lower porosity), the use of silica fume or fly ash possolans, or by using surface coatings.8
Concrete is also subject to degradation from an alkali silicate reaction (ASR) if there is incorrect selection of aggregate. Some silicate minerals used as aggregate will react with sodium or potassium alkali in high pH conditions to form a higher-volume product which can put expansive stresses on the concrete and cause cracking. While the alkali metals sodium and potassium that are present in cement are typically low, external sources of alkali like deicing salts or sea water can lead to ASR. Reducing the ingress of external alkalis through less permeable concrete (lower w/c) or using coatings or sealers can reduce the potential for the ASR.3
Chloride ions can also be a significant problem in steel reinforced concrete. Steel reinforcement rods in wet concrete quickly form a protective rust layer at the surface which protect the rods from further degradation. The chloride ions destroy the protective Fe2O3 passivating layer on steel and, if there is water and air present in the concrete pores, then an electrochemical current is set up and rust is formed at the anode. In addition to causing pitting in the steel, the rust has a larger volume than the steel and it gives expansive pressure on the surrounding concrete and can cause cracking of the concrete.5 Decreasing the porosity with a lower water to cement ratio and the use of fly ash can mitigate chloride attack, however these preventative measures are often used in conjunction with concrete coatings or coatings for the steel surface.
If deicing salts are used, they can collect in the voids at the surface of the concrete. The crystallization of the salts can put stress on the surface layers of concrete and cause scaling. This is worse in concrete exposed to repeated wet/dry cycles. Coatings which protect the surface from moisture can prevent this type of degradation.4
Concrete can also degrade on exposure to acid. Acids can dissolve both hydrated and unhydrated cement compounds and can dissolve calcium carbonate (calcareous) based aggregate. Acid rain (pH 4.5-5) can lead to slight etching of the surface but does not significantly affect performance. Reducing the porosity of the concrete with coatings can reduce the degradation.4
Another common concrete failure is caused by freeze thaw cycles. The freezing of water in the pores creates stresses in the concrete which can lead to spalling. It is common to use air entraining additives when mixing wet concrete to prevent damage from freeze thaw cycles. The entrained air pockets in the dried concrete give water room to expand and prevent damage from freezing. Another way to help prevent damage is to reduce the porosity which can help prevent the ingress of water. The porosity can be reduced by reducing the water to cement ratio as much as possible or by using sealers.
Sealers
Polymeric sealers can help prevent the chemical or physical degradation of concrete. Sealers are typically described as penetrating or film forming and are differentiated from cure and seal coatings since they are applied after the concrete has cured for 28 days.
Silanes or siloxanes are penetrating sealers. They do help seal the surface against liquid water and stains but they do not prevent water vapor loss from the concrete during cure. The silanes have a low molecular weight while the siloxanes are prereacted and have an EO chain giving higher molecular weight. Because of the lower molecular weight, the silanes can give more penetration into the pores than the silanes. Since they are beneath the surface, they are more abrasion resistant than the aqueous sealers which form a film on the surface.
Aqueous emulsions used as sealers can also help prevent ingress of salts and moisture which can lead to degradation. These are typically acrylic or styrene acrylic and have good resistance to liquid water but are breathable and allow vapor to pass through the film. In addition to water resistance, water whitening resistance is also needed to avoid discoloration if exposed to puddled water. Polymers need to be designed to be blush resistant by keeping the particle size small, using special polymerizable surfactants and keeping the level of other hydrophilic materials (such as surfactants) low. These emulsion sealers are typically designed to be applied at low dry film thicknesses and they are typically not high gloss finishes – usually satin or semigloss finishes. Since the films are relatively thin and largely on the surface of the concrete, they usually need to be reapplied every other year to maintain protection.
Solvent-based acrylics are also commonly used as sealers and are usually applied after the concrete has aged for a month. These sealers are very durable because of the acrylic backbone, but still low enough molecular weight to penetrate into the concrete pores and provide a darkening of the surface (called a wet look) which can be desirable. They are applied at higher film thicknesses than the acrylic emulsions and can give a high gloss when dried. These acrylics can be formulated in exempt solvents like acetone, however the rapid dry speed and odor can present a significant issue during application.
Two part epoxies are used to seal concrete in environments where a high level of chemical resistance is needed. These can be cured with polyamines, polyamides or polysulfides, although the polyamide-cured epoxies typically have lower chemical resistance. The epoxies have good adhesion to the concrete and can be formulated to be relatively oil tolerant for use on garage floors, or designed to cure at low temperatures. Epoxies can chalk on exterior exposure, however, and they are not usually used for outdoor applications.
In the case of chloride attack on the reinforcing steel, it is also possible to reduce the effect by applying a thin overlayment of polymer modified cement to reduce the surface porosity of concrete. The polymeric admixes function by reducing the amount of water necessary for a given concrete fluidity resulting in a lower water to cement ratio. In addition, as the concrete dries the polymer forms films in the pores and blocks the capillaries. The slower cure of the concrete and the dried polymer in the matrix results in reduced porosity.6 While the reduced porosity of the overlayment helps reduce the penetration of water carrying chloride ions, the polymer can also improve adhesion to the concrete substrate and can improve durability by increasing the tensile strength, resistance to abrasion and dusting.
Summary
In order for the complex chemical reactions in concrete to occur, water is required. To build optimum strength and avoid cracking at the surface, it is also necessary to keep concrete moist for several days after being poured. Polymeric coatings and cure and seal coatings can help develop the strength of concrete by reducing the porosity at the surface and keeping the water in the concrete longer. Cured concrete is also subject to chemical attack or physical degradation by freeze thaw cycles. Sealers or polymer-modified overlayments applied after cure can help protect concrete from these forms of degradation.
References
1. Thomas, J.; Jennings, H. (n.d.). Science of Concrete. Retrieved June 28, 2016, from http://iti.northwestern.edu/cement/index.html
2. Thomas, M. (2007). Optimizing the use of Fly Ash in Concrete. Retrieved July 15, 2016, from http://www.cement.org/docs/default-source/fc_concrete_technology/is548-optimizing-the-use-of-fly-ash-concrete.pdf
3. Farny, J.; Kerkhoff, B. (2007). Diagnosis and Control of Alkali-Aggregate Reactions in Concrete. Retrieved June 14, 2016, from http://www.cement.org/docs/default-source/fc_concrete_technology/is413-02---diagnosis-and-control-of-alkali-aggregate-reactions-in-concrete.pdf?sfvrsn=2
4. Kerkhoff, B. (2007). Effects of Substances on Concrete and Guide to Protective Treatments [Pamphlet]. Skokie, IL: Portland Cement Association.
5. Neville, A. (1995). Chloride attack of reinforced concrete: An overview. Materials and Structures, 28, 63-70.
6. ATS 11 - Polymer Dispersion Admixtures. (2016, April 21). Retrieved June 14, 2016, from http://www.admixtures.org.uk/download/ats-11-polymer-dispersion-admixtures/
7. Duvallet, T. (2014). Influence Of Ferrite Phase In Alite Calcium Sulfoaluminate Cements (Published doctoral dissertation, 2014). University of Kentucky. Retrieved August 8, 2016, from http://uknowledge.uky.edu/cme_etds/27/
8. Kurtis, K. (2015) Portland Cement Hydration. Retrieved July 7, 2016 from http://people.ce.gatech.edu/~kk92/hyd07.pdf
9. Stark, D. (2002) Performance of Concrete in Sulfate Environments, [Pamphlet]. Skokie, IL: Portland Cement Association.