Richard Najdusak and Dr. Franjo Gol 06.08.16
Being a formulator is becoming much more difficult in light of increased regulatory pressures on common raw materials.
Formulators who develop coatings based on oxidatively cured (alkyd) binders face major regulatory obstacles for two key ingredients: methyl ethyl ketoxime (MEKO) anti-skinning additive and cobalt-based driers. MEKO’s toxicity makes it difficult to meet safe exposure limits due to its volatility. Cobalt carboxylates are being pressured to be classified as carcinogens. In this paper we discuss potential non-toxic alternatives to both of these materials with supporting test results.
Binders that Cure via Oxidation
Drying oils and alkyd resins have many advantages over other binders used in the coatings industry. They are bio-renewable, inexpensive and extremely stable. Alkyds can be used to formulate coatings which have long open times for easier application, yield relatively hard films for kitchens and baths, require minimal surface preparation and provide good stain blocking properties, particularly over water-based stains, inks and markers. Water-based versions of these binders have been developed to meet VOC regulations, have low odor and water clean-up. Alkyd resins are used in architectural, light duty industrial, wood care, printing inks and automotive aftermarket coatings.
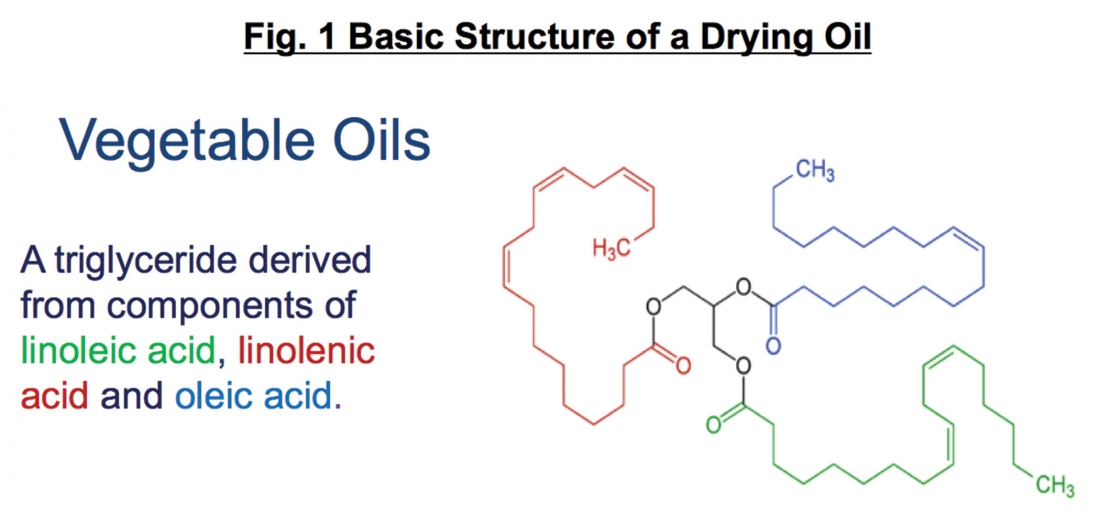
Drying oils, alkyd resins and epoxy esters have certain things in common. They all contain some degree of unsaturation (double bonds) and hydroxyl/carboxyl functionality which are used to promote curing. In the following example of a common vegetable oil, the triglyceride contains a range of organic acids which have various degrees of unsaturation (Figure 1). Those with more potential crosslinking (unsaturation) sites, such as linolenic acid, can be expected to dry faster and yield harder finishes, while binders with high levels of oleic acid have few unsaturation sites and will yield softer, slow drying films.
The Need for Driers
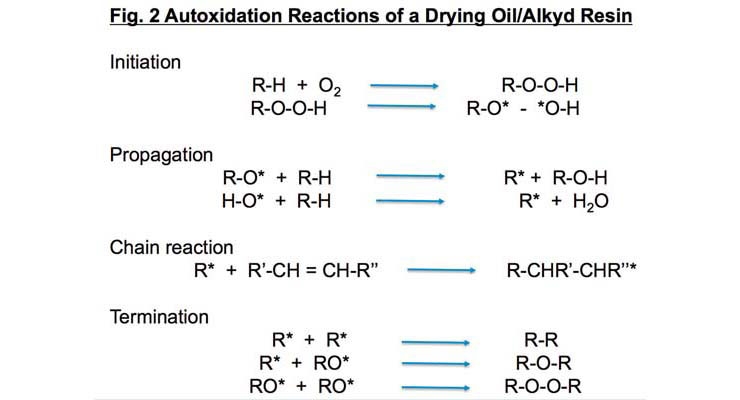
Atmospheric oxygen reacts spontaneously with the unsaturated fatty acid components of oils and alkyd resins generating free radical reactions to eventually form films (Figure 2). This process is called autoxidation. However, if unaided, the reaction rates are very slow.
Surface (or oxidative) driers catalyze reactions by: deactivating the natural antioxidants found in drying oils by forming hydroperoxides; accelerating oxygen absorption and peroxide formation; reacting with oxygen or hydroperoxides to form complexes that catalyze oxidation reactions; and acting as oxygen carriers. Surface driers are also susceptible to redox reactions which can decompose hydroperoxides and promote crosslinking.
Metal Catalyzed Reactions
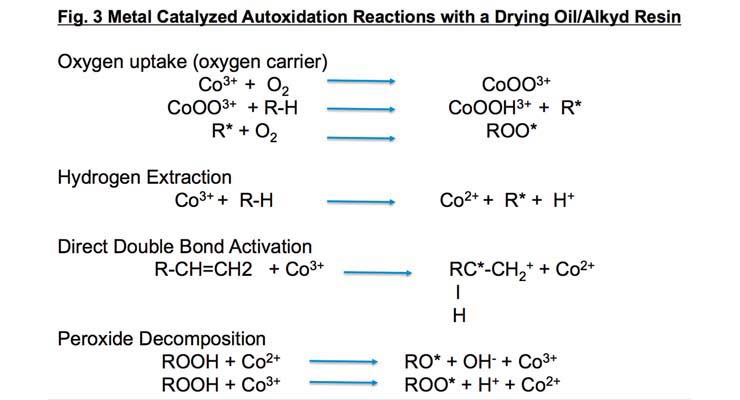
Adding metal catalysts greatly improves the oxidation rate of the binder (Figure 3). When a metal catalyst is present, the activation energy for oxygen uptake is only 10% of the amount needed when none is present. Peroxide formation also proceeds more rapidly. Metal catalysts aid in the decomposition of the peroxides which accelerates free radical formation to promote crosslinking.
Another advantage is that the lower valence state of the metal is regenerated making most oxidative metal carboxylates true catalysts which, unfortunately, is not always a good thing as will be discussed.
Metals Capable of Use in Oxidative (Surface) Driers
There are only five metals that promote oxidative or surface curing in some form. Cobalt is the most common oxidative drier and has been commercially available since the early 1900s. It exists in the two valance states noted in Figure 3 which helps it form coordination complexes with oxygen and organic molecules. The activity of cobalt driers is inhibited at lower temperatures and its use in high humidity drying conditions may promote film wrinkling.
Manganese is more efficient than cobalt at low temperatures and also when used in baking finishes, but not at ambient temperatures. It is mainly used in drying oils in combination with cobalt. As a cobalt replacement, manganese requires the addition of a drier accelerator like 1,10-phenanthroline or 2,2’-bipyridyl chelating additives to approach the activity level of cobalt. Films may develop a brown colored hue on ageing due to the color of the manganese drier.
Iron has no catalytic activity at room temperature in air drying finishes but it is more active than cobalt at elevated temperatures (130 °C). The dark red color of iron driers restricts its usage in coatings to dark colors and red coatings.
Vanadium is efficient at lower temperatures but has a tendency to impart a greenish stain to films. Unlike cobalt, vanadium is more stable in its higher valence state which inhibits its role in oxidative curing and complex formation. Higher levels are generally needed.
Cerium can promote oxidative curing at elevated temperatures but is primarily used as a through or coordinate drier.
Iron-Complex technology - A new unique organic complex with iron has yielded an oxidative drier which magnifies the drying activity of iron so that it is effective over a wide range of temperatures and drying conditions. This new iron-complex technology is more active than cobalt, has a very light color and promotes oxidative curing both at the surface and throughout the film in many binders.
Potential Cobalt Re-Classification
Cobalt driers have been a regulatory concern for many years. Reproductive toxicity and the long term adverse effects on aquatic organisms have been well known and documented. As described above, cobalt driers are true catalysts in the oxidation process of air drying binders in that they do not become part of the molecular structure. Cobalt driers do not crosslink into the film and also have some water solubility so they will eventually leach into the ground water supply when the films come in contact with water. There is also the growing concern that cobalt (as cobalt 2-ethylhexanoate, at least) will be classified as a carcinogen. Most European paint manufacturers have already replaced cobalt in their formulations in anticipation of one or all of these concerns being realized.
Cobalt Replacement Strategies
Few viable options exist to replace cobalt in alkyd and other air drying systems. As noted above, only manganese and iron complexes can approach cobalt’s activity level. Conventional iron, vanadium and cerium carboxylate driers are not effective replacements. “Accelerated” manganese driers improve the activity somewhat but may affect the color of light tinted coatings over time and generate brittle films. However, a new iron-complex drier has been developed to equal or exceed the drying performance of cobalt and at reduced levels. The catalytic reaction mechanisms of cobalt and the iron-complex driers have been shown to be the same, however the efficiency of the iron-complex drier is so high that less oxygen is needed to promote oxidative cure throughout the film, not just on the surface as do the cobalt carboxylates.
Iron Complex Drier - FeLT®
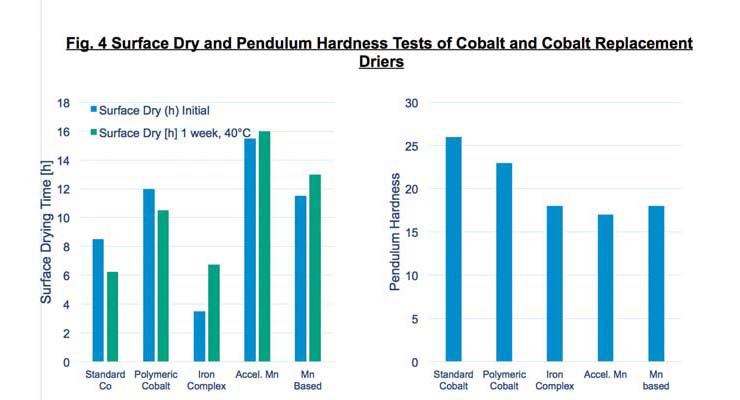
The new iron-complex drier is an effective oxidative drier for a wide variety of binder systems, particularly alkyd emulsions. Figure 4 shows the surface dry time and Pendulum hardness test results of several cobalt and manganese-based driers compared to the iron-complex drier in a long-oil alkyd resin.
The test systems were evaluated for dry initially and after one week exposure at 40 ºC. Pendulum hardness tests were run after one week cure. Note that the new iron-complex developed faster surface drying at ambient temperatures than any of the other products tested.
A major advantage of the iron-complex drier is its ability to promote improved oxidative cure under marginal drying conditions which typically affect conventional surface driers. In a white, low-VOC high-solids alkyd-based formula, the iron-complex drier was not only more effective than cobalt and accelerated manganese-based driers under standard drying conditions, but also was virtually unaffected in low temperature/high humidity environments where the others experienced decreased activity (Figure 5).
A major concern about coatings based on alkyd and drying oil films is their tendency to yellow in the absence of UV light.
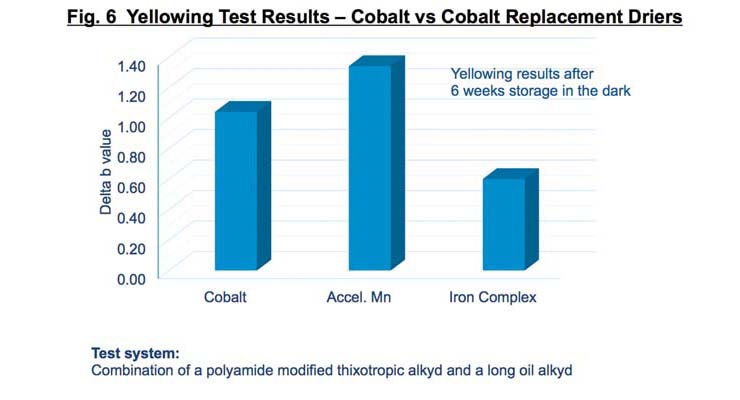
Typically a cocktail of through driers is necessary to reduce yellowing by forming coordinate bonds with the low-molecular-weight chromophores that are generated during the curing process. The additional through drying property of the iron-complex drier greatly reduces the film’s tendency to yellow as seen in Figure 6.
Reduced yellowing is believed to be due to the increased level of deep film oxidative crosslinking from the higher activity of the iron-complex drier. However, as with cobalt, coordinative driers like zirconium may still be needed to promote faster hardness values.
Summary of Cobalt Replacement Driers
No other metal carboxylate drier can approach the oxidative activity of cobalt. However, the iron-complex drier is a sustainable alternative to cobalt driers and is not carboxylate-based. This product is not CMR classified, can provide equal or superior surface dry times to cobalt at low levels under standard drying conditions, has improved efficiency in promoting surface curing under low temperature/high humidity conditions and, due to its additional through drying properties, can reduce long-term film yellowing.
Using Anti-Skinning Additives in Paints
By adding driers to improve coating dry times, we also increase the tendency of the coating to cure prematurely whenever it comes in contact with air. This includes during the manufacturing, filtering and filling processes. Adding driers also means that less oxygen is needed to initiate surface curing which can affect long-term storage in the can. The coatings industry developed additives to delay the curing process to prevent skin formation. The two main methods to prevent skinning are: (1) inhibit the catalytic activity of the metal carboxylate and (2) absorb the free radicals during autoxidation to prevent crosslinking.
The first method involves using a volatile material to form temporary complexes with the metal in the drier to block its catalytic activity. The inhibitor evaporates quickly during film application freeing the metal to function. The most common chemical used for this is methyl ethyl ketoxime (MEKO), or ketoximes in general. They are especially effective with cobalt, however, their high volatility makes them a health concern. The major toxicity is to erythrocytes and the hematopoietic system. Ketoximes cause similar lesions in the olfactory epithelium. Hyperplasia of the urinary bladder transitional epithelium was observed in mice exposed to MEKO but not with a similar chemical, cyclohexanone oxime. For MEKO, the “no observed-adverse-effect level” (NOAEL) for erythrotoxicity is 312 ppm in the drinking water for rats and 2,500 ppm for mice (see National Toxicology Program, Toxicity Report Series, Number 51, July 1999, NIH Publication 99-3947).
Figure 7 shows part of the SDS for MEKO with regard to Health Hazards (2.1) and Label Elements (2.2).
The second method to prevent skinning involves using antioxidants to absorb free radicals in the coating. However, these can severely inhibit oxidation and affect curing and film appearance if excessive amounts are used. The coating can only cure after the antioxidant has been consumed (with free radicals), leaves the film through evaporation or is driven off during the bake cycle (if part of the curing system). These consist of substituted phenolics, quinones, aromatic amines, sterically-hindered aliphatic amines and hydroxyl amines.
The most preferred anti-skinning additive would be non-toxic, have no regulatory concerns, have no effect on the cured film and still prevent skin formation.
MEKO Replacement
Most antioxidants are unsuitable for architectural and industrial applications due to their side effects. However, a proprietary aminic compound has been developed which is non-toxic, volatile and consumes free radicals. This “advanced antioxidant” provides a controlled retardation of surface dry resulting in improved through dry without affecting the film hardness. Another advantage is that its activity is independent of the drier metal. This new product is more volatile than other types of antioxidants, but not as volatile as MEKO. It can be used in clear and pigmented systems and has been successfully marketed since 2002.
Aminic Complex
The health hazards associated with this advanced antioxidant are minimal compared to MEKO as noted on its current SDS (Figure 8).
Unlike MEKO, the proprietary aminic complex is not harmful to humans and only in concentrated form can be harmful to aquatic life if discharged directly into watercourses.
Comparative Testing to MEKO
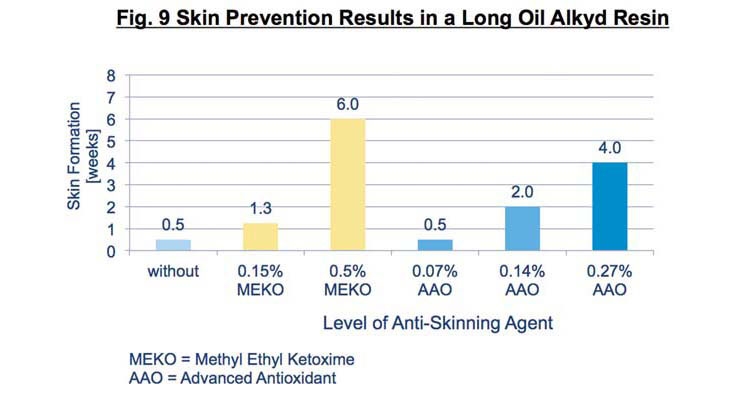
These “advanced antioxidants” were evaluated for in-can preservation, film surface drying and film hardness development vs MEKO. MEKO is completely volatile and does not interfere with curing while all antioxidants remain, at least for a period of time, in the film to function properly. In these tests, we chose a simple, clear alkyd formula based on a long-oil alkyd resin and used a drier package consisting of 0.04% cobalt and 0.1% calcium metal based on resin solids. Results are shown in Figure 9.
Although not as effective as MEKO with this drier package, the advanced antioxidants at low levels provide excellent anti-skinning properties to this test system.
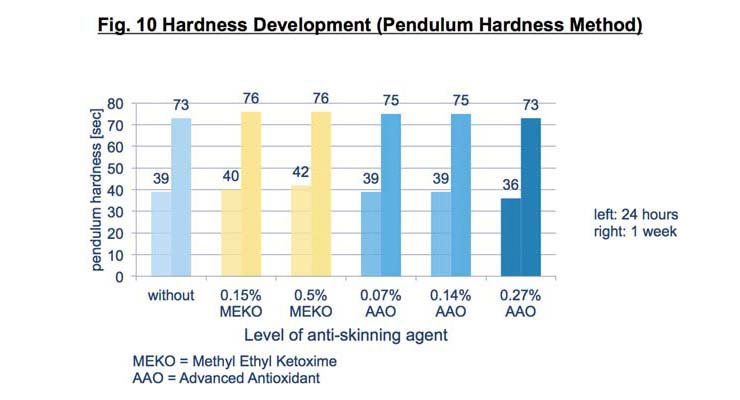
The overall hardness and integrity of the dried film was not compromised initially or after one week using the advanced antioxidant complex at any of the levels tested (Figure 10). This confirms that it does not remain in the film to interfere with crosslinking and final cure.
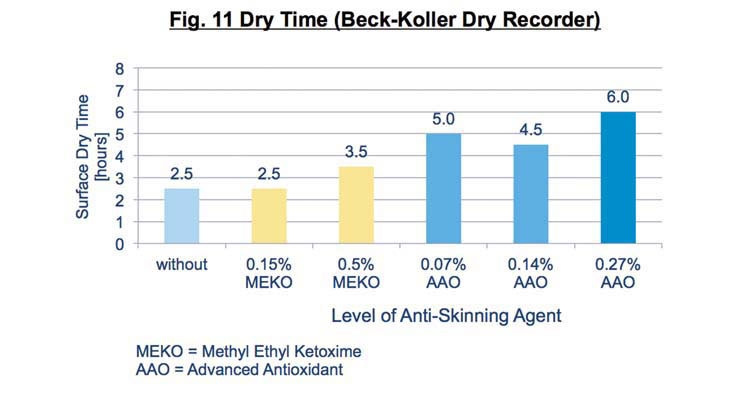
There is a slight loss of surface dry using the advanced antioxidant complex which is due to its lower volatility compared to MEKO and the different mechanism for inhibiting oxidation (Figure 11). Excessive amounts should be avoided for these reasons.
Summary – MEKO Replacement
These less toxic alternatives to phenolic and oximic antioxidants require less than half the level of MEKO in most coatings systems to provide effective anti-skinning properties. They perform independently of the oxidative metal drier and are recommended in cobalt replacement strategies. These “advanced antioxidants” are suitable for clear and pigmented coatings with no discoloration. However, the optimum level should be experimentally determined as excessive amounts may lead to longer dry times due to their volatility being less than that of MEKO.
Conclusions
Additives manufacturers have developed less toxic alternatives to cobalt and MEKO which are both more effective and efficient.
Cobalt Replacement – Iron-Complex technology is an effective and sustainable alternative to cobalt driers with the following advantages: no CMR classification issues; improved film curing properties under adverse conditions (high humidity/low temperature) compared to cobalt; and dried films develop less yellowing compared to films cured using cobalt driers.
MEKO Replacement – Effective, less toxic, aminic-based anti-skinning alternatives are available with minimal loss of properties if used at the appropriate levels.
Formulators who develop coatings based on oxidatively cured (alkyd) binders face major regulatory obstacles for two key ingredients: methyl ethyl ketoxime (MEKO) anti-skinning additive and cobalt-based driers. MEKO’s toxicity makes it difficult to meet safe exposure limits due to its volatility. Cobalt carboxylates are being pressured to be classified as carcinogens. In this paper we discuss potential non-toxic alternatives to both of these materials with supporting test results.
Binders that Cure via Oxidation
Drying oils and alkyd resins have many advantages over other binders used in the coatings industry. They are bio-renewable, inexpensive and extremely stable. Alkyds can be used to formulate coatings which have long open times for easier application, yield relatively hard films for kitchens and baths, require minimal surface preparation and provide good stain blocking properties, particularly over water-based stains, inks and markers. Water-based versions of these binders have been developed to meet VOC regulations, have low odor and water clean-up. Alkyd resins are used in architectural, light duty industrial, wood care, printing inks and automotive aftermarket coatings.
Drying oils, alkyd resins and epoxy esters have certain things in common. They all contain some degree of unsaturation (double bonds) and hydroxyl/carboxyl functionality which are used to promote curing. In the following example of a common vegetable oil, the triglyceride contains a range of organic acids which have various degrees of unsaturation (Figure 1). Those with more potential crosslinking (unsaturation) sites, such as linolenic acid, can be expected to dry faster and yield harder finishes, while binders with high levels of oleic acid have few unsaturation sites and will yield softer, slow drying films.
The Need for Driers
Atmospheric oxygen reacts spontaneously with the unsaturated fatty acid components of oils and alkyd resins generating free radical reactions to eventually form films (Figure 2). This process is called autoxidation. However, if unaided, the reaction rates are very slow.
Surface (or oxidative) driers catalyze reactions by: deactivating the natural antioxidants found in drying oils by forming hydroperoxides; accelerating oxygen absorption and peroxide formation; reacting with oxygen or hydroperoxides to form complexes that catalyze oxidation reactions; and acting as oxygen carriers. Surface driers are also susceptible to redox reactions which can decompose hydroperoxides and promote crosslinking.
Metal Catalyzed Reactions
Adding metal catalysts greatly improves the oxidation rate of the binder (Figure 3). When a metal catalyst is present, the activation energy for oxygen uptake is only 10% of the amount needed when none is present. Peroxide formation also proceeds more rapidly. Metal catalysts aid in the decomposition of the peroxides which accelerates free radical formation to promote crosslinking.
Another advantage is that the lower valence state of the metal is regenerated making most oxidative metal carboxylates true catalysts which, unfortunately, is not always a good thing as will be discussed.
Metals Capable of Use in Oxidative (Surface) Driers
There are only five metals that promote oxidative or surface curing in some form. Cobalt is the most common oxidative drier and has been commercially available since the early 1900s. It exists in the two valance states noted in Figure 3 which helps it form coordination complexes with oxygen and organic molecules. The activity of cobalt driers is inhibited at lower temperatures and its use in high humidity drying conditions may promote film wrinkling.
Manganese is more efficient than cobalt at low temperatures and also when used in baking finishes, but not at ambient temperatures. It is mainly used in drying oils in combination with cobalt. As a cobalt replacement, manganese requires the addition of a drier accelerator like 1,10-phenanthroline or 2,2’-bipyridyl chelating additives to approach the activity level of cobalt. Films may develop a brown colored hue on ageing due to the color of the manganese drier.
Iron has no catalytic activity at room temperature in air drying finishes but it is more active than cobalt at elevated temperatures (130 °C). The dark red color of iron driers restricts its usage in coatings to dark colors and red coatings.
Vanadium is efficient at lower temperatures but has a tendency to impart a greenish stain to films. Unlike cobalt, vanadium is more stable in its higher valence state which inhibits its role in oxidative curing and complex formation. Higher levels are generally needed.
Cerium can promote oxidative curing at elevated temperatures but is primarily used as a through or coordinate drier.
Iron-Complex technology - A new unique organic complex with iron has yielded an oxidative drier which magnifies the drying activity of iron so that it is effective over a wide range of temperatures and drying conditions. This new iron-complex technology is more active than cobalt, has a very light color and promotes oxidative curing both at the surface and throughout the film in many binders.
Potential Cobalt Re-Classification
Cobalt driers have been a regulatory concern for many years. Reproductive toxicity and the long term adverse effects on aquatic organisms have been well known and documented. As described above, cobalt driers are true catalysts in the oxidation process of air drying binders in that they do not become part of the molecular structure. Cobalt driers do not crosslink into the film and also have some water solubility so they will eventually leach into the ground water supply when the films come in contact with water. There is also the growing concern that cobalt (as cobalt 2-ethylhexanoate, at least) will be classified as a carcinogen. Most European paint manufacturers have already replaced cobalt in their formulations in anticipation of one or all of these concerns being realized.
Cobalt Replacement Strategies
Few viable options exist to replace cobalt in alkyd and other air drying systems. As noted above, only manganese and iron complexes can approach cobalt’s activity level. Conventional iron, vanadium and cerium carboxylate driers are not effective replacements. “Accelerated” manganese driers improve the activity somewhat but may affect the color of light tinted coatings over time and generate brittle films. However, a new iron-complex drier has been developed to equal or exceed the drying performance of cobalt and at reduced levels. The catalytic reaction mechanisms of cobalt and the iron-complex driers have been shown to be the same, however the efficiency of the iron-complex drier is so high that less oxygen is needed to promote oxidative cure throughout the film, not just on the surface as do the cobalt carboxylates.
Iron Complex Drier - FeLT®
The new iron-complex drier is an effective oxidative drier for a wide variety of binder systems, particularly alkyd emulsions. Figure 4 shows the surface dry time and Pendulum hardness test results of several cobalt and manganese-based driers compared to the iron-complex drier in a long-oil alkyd resin.
The test systems were evaluated for dry initially and after one week exposure at 40 ºC. Pendulum hardness tests were run after one week cure. Note that the new iron-complex developed faster surface drying at ambient temperatures than any of the other products tested.
A major advantage of the iron-complex drier is its ability to promote improved oxidative cure under marginal drying conditions which typically affect conventional surface driers. In a white, low-VOC high-solids alkyd-based formula, the iron-complex drier was not only more effective than cobalt and accelerated manganese-based driers under standard drying conditions, but also was virtually unaffected in low temperature/high humidity environments where the others experienced decreased activity (Figure 5).
A major concern about coatings based on alkyd and drying oil films is their tendency to yellow in the absence of UV light.
Typically a cocktail of through driers is necessary to reduce yellowing by forming coordinate bonds with the low-molecular-weight chromophores that are generated during the curing process. The additional through drying property of the iron-complex drier greatly reduces the film’s tendency to yellow as seen in Figure 6.
Reduced yellowing is believed to be due to the increased level of deep film oxidative crosslinking from the higher activity of the iron-complex drier. However, as with cobalt, coordinative driers like zirconium may still be needed to promote faster hardness values.
Summary of Cobalt Replacement Driers
No other metal carboxylate drier can approach the oxidative activity of cobalt. However, the iron-complex drier is a sustainable alternative to cobalt driers and is not carboxylate-based. This product is not CMR classified, can provide equal or superior surface dry times to cobalt at low levels under standard drying conditions, has improved efficiency in promoting surface curing under low temperature/high humidity conditions and, due to its additional through drying properties, can reduce long-term film yellowing.
Using Anti-Skinning Additives in Paints
By adding driers to improve coating dry times, we also increase the tendency of the coating to cure prematurely whenever it comes in contact with air. This includes during the manufacturing, filtering and filling processes. Adding driers also means that less oxygen is needed to initiate surface curing which can affect long-term storage in the can. The coatings industry developed additives to delay the curing process to prevent skin formation. The two main methods to prevent skinning are: (1) inhibit the catalytic activity of the metal carboxylate and (2) absorb the free radicals during autoxidation to prevent crosslinking.
The first method involves using a volatile material to form temporary complexes with the metal in the drier to block its catalytic activity. The inhibitor evaporates quickly during film application freeing the metal to function. The most common chemical used for this is methyl ethyl ketoxime (MEKO), or ketoximes in general. They are especially effective with cobalt, however, their high volatility makes them a health concern. The major toxicity is to erythrocytes and the hematopoietic system. Ketoximes cause similar lesions in the olfactory epithelium. Hyperplasia of the urinary bladder transitional epithelium was observed in mice exposed to MEKO but not with a similar chemical, cyclohexanone oxime. For MEKO, the “no observed-adverse-effect level” (NOAEL) for erythrotoxicity is 312 ppm in the drinking water for rats and 2,500 ppm for mice (see National Toxicology Program, Toxicity Report Series, Number 51, July 1999, NIH Publication 99-3947).
Figure 7 shows part of the SDS for MEKO with regard to Health Hazards (2.1) and Label Elements (2.2).
The second method to prevent skinning involves using antioxidants to absorb free radicals in the coating. However, these can severely inhibit oxidation and affect curing and film appearance if excessive amounts are used. The coating can only cure after the antioxidant has been consumed (with free radicals), leaves the film through evaporation or is driven off during the bake cycle (if part of the curing system). These consist of substituted phenolics, quinones, aromatic amines, sterically-hindered aliphatic amines and hydroxyl amines.
The most preferred anti-skinning additive would be non-toxic, have no regulatory concerns, have no effect on the cured film and still prevent skin formation.
MEKO Replacement
Most antioxidants are unsuitable for architectural and industrial applications due to their side effects. However, a proprietary aminic compound has been developed which is non-toxic, volatile and consumes free radicals. This “advanced antioxidant” provides a controlled retardation of surface dry resulting in improved through dry without affecting the film hardness. Another advantage is that its activity is independent of the drier metal. This new product is more volatile than other types of antioxidants, but not as volatile as MEKO. It can be used in clear and pigmented systems and has been successfully marketed since 2002.
Aminic Complex
The health hazards associated with this advanced antioxidant are minimal compared to MEKO as noted on its current SDS (Figure 8).
Unlike MEKO, the proprietary aminic complex is not harmful to humans and only in concentrated form can be harmful to aquatic life if discharged directly into watercourses.
Comparative Testing to MEKO
These “advanced antioxidants” were evaluated for in-can preservation, film surface drying and film hardness development vs MEKO. MEKO is completely volatile and does not interfere with curing while all antioxidants remain, at least for a period of time, in the film to function properly. In these tests, we chose a simple, clear alkyd formula based on a long-oil alkyd resin and used a drier package consisting of 0.04% cobalt and 0.1% calcium metal based on resin solids. Results are shown in Figure 9.
Although not as effective as MEKO with this drier package, the advanced antioxidants at low levels provide excellent anti-skinning properties to this test system.
The overall hardness and integrity of the dried film was not compromised initially or after one week using the advanced antioxidant complex at any of the levels tested (Figure 10). This confirms that it does not remain in the film to interfere with crosslinking and final cure.
There is a slight loss of surface dry using the advanced antioxidant complex which is due to its lower volatility compared to MEKO and the different mechanism for inhibiting oxidation (Figure 11). Excessive amounts should be avoided for these reasons.
Summary – MEKO Replacement
These less toxic alternatives to phenolic and oximic antioxidants require less than half the level of MEKO in most coatings systems to provide effective anti-skinning properties. They perform independently of the oxidative metal drier and are recommended in cobalt replacement strategies. These “advanced antioxidants” are suitable for clear and pigmented coatings with no discoloration. However, the optimum level should be experimentally determined as excessive amounts may lead to longer dry times due to their volatility being less than that of MEKO.
Conclusions
Additives manufacturers have developed less toxic alternatives to cobalt and MEKO which are both more effective and efficient.
Cobalt Replacement – Iron-Complex technology is an effective and sustainable alternative to cobalt driers with the following advantages: no CMR classification issues; improved film curing properties under adverse conditions (high humidity/low temperature) compared to cobalt; and dried films develop less yellowing compared to films cured using cobalt driers.
MEKO Replacement – Effective, less toxic, aminic-based anti-skinning alternatives are available with minimal loss of properties if used at the appropriate levels.