Steven Y. Chan, PhD., Nathan Noyes, Croda, New Castle, DE08.10.18
Introduction
As consumer awareness of the products and chemicals they use rises, many chemical companies are also striving to become more environmentally responsible. The trend in the coatings industry is toward low/zero VOC products, and many innovations in coatings technology are based on sustainable or green chemistries. The market increasingly demands the use of renewable ingredients to reduce the carbon footprint, and many chemical and coating manufacturers are striving to become more environmentally responsible. This trend is driven by a number of factors, including evolving consumer perceptions and awareness regarding the products and chemicals they use, regulatory/certification bodies and programs such as USDA BioPreferred®, and the growing implementation of corporate sustainability initiatives. Bio-based ethylene oxide (EO) will meet this demand by enabling the synthesis of various ethoxylated surfactants and emulsifiers which are 100% bio-based.
Ethoxylation is a common process used to generate a range of products for emulsification and wetting, including ethoxylated alcohols, carboxylic acids and esters. While the hydrophobic portions of many of these surfactants are already naturally sourced from plant oils, only petrochemical-derived EO has been available in North America until this time. With the production of bio-based EO in the near future, ethoxylated products can now be produced from 100% bio-based content, allowing customers to choose fully renewable products without sacrificing performance. In addition, by incorporation into synthetic base materials, the bio-based content can be significantly increased, allowing formulators to meet challenging new targets.
Surfactant Fundamentals
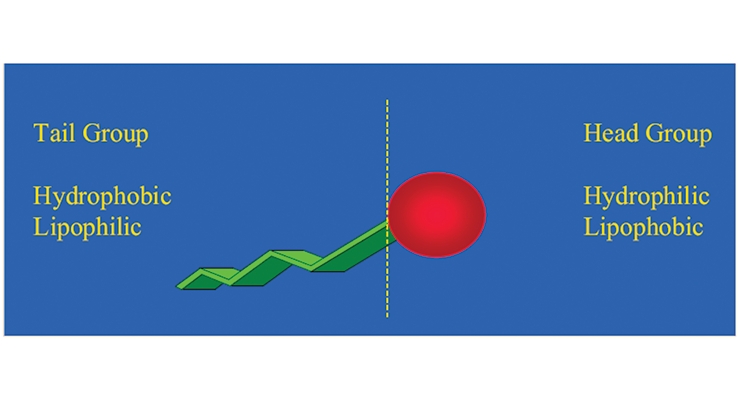
In coatings, especially waterborne, surfactants are used as resin emulsifiers and stabilizers, pigment dispersants, grinding aids, compatibilizers, wetting agents and in defoamer and other additive packages. The word “surfactant” is the contraction of a surface active agent. Surfactants are usually organic compounds that are amphiphilic in nature, containing a lipophilic (oil like) tail and a hydrophilic (water like) head group. The general structure is illustrated in Figure 1. This unique structure enables surfactants to adsorb onto the surfaces and interfaces in a system at low concentration. The chemistry and types of surfactants are well known.1,2
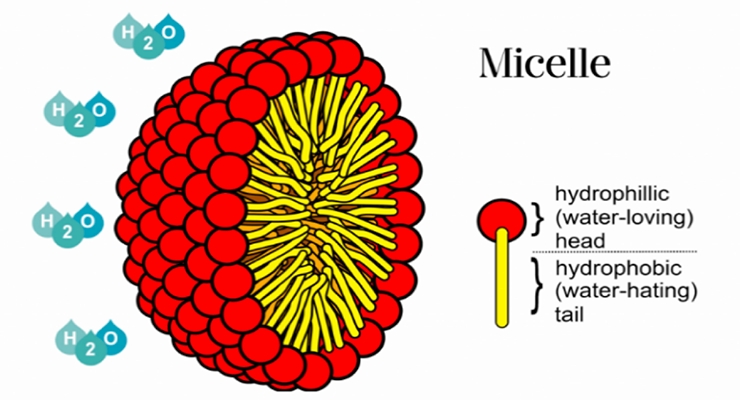
When adsorbed, surfactants will lower the surface or interfacial tension. In the bulk aqueous phase, surfactants tend not to be fully soluble but rather form aggregates as micelles³ as illustrated in Figure 2. Beside spherical, other types such as cylindrical or bilayer type micelles can be formed depending on the chemistry and structure of the surfactant. The behaviors of surfactants in liquid media contribute to their great importance and tunability in applications like foaming, emulsifying, dispersing, and wetting.
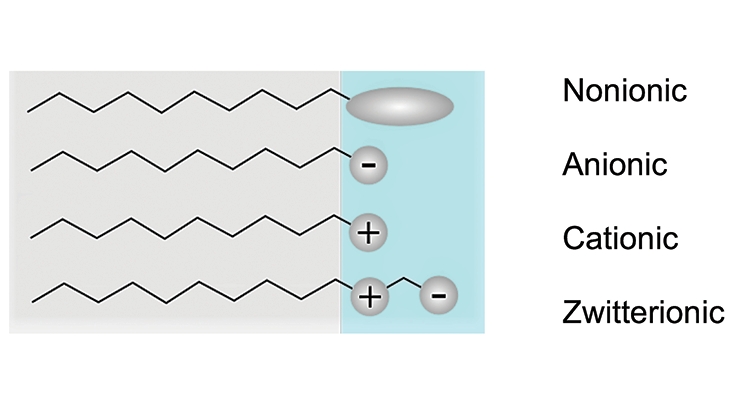
Most common surfactants contain a hydrocarbon “tail” which can be linear, branched or aromatic. Other types of surfactants with tails based on siloxane and fluorocarbons are also used, but to a lesser extent in the industry. The nature of the hydrophilic head group determines the type of the surfactant: nonionic, anionic, cationic, or amphoteric, as illustrated in Figure 3. Nonionic surfactants have no charged group in the head portion of the molecule. In structures where the head carries a negative or positive charge, it is called anionic or cationic respectively. Zwitterionic is a term for surfactants with a head group consisting of two oppositely charged species.
Common anionic surfactants are sodium, potassium or ammonium salts of alkyl sulfate, sulfonate, phosphate, and sulfosuccinate.
The most common cationic surfactants are the quaternary ammonium salts, typically bromide or chloride. For zwitterionic surfactants, amines or quaternary ammonium cations make up the cationic part and the anionic portion is commonly a sulfonate or carboxylate. Alkyl betaines, and amine oxides are pH sensitive zwitterionics, displaying anionic or nonionic properties above the isoelectric point, and cationic below the isoelectric point. Sulfobetaines and sultaines are not pH sensitive.
Nonionic surfactants have several advantages over ionic types for many applications. They tend to be more compatible in a broader range of systems, which makes them generally easier to formulate with. They have much better resistance to hard water, polyvalent metallic cations, and electrolytes. Polyoxyethylenated organic compounds dominate the market of nonionic surfactants. The hydrophobic tail group could be alkylphenols, straight or branched alcohols, polyoxypropylene glycols, sorbitans or sorbitan esters, and glycol esters or fatty acids. Other organic nonionics include tertiary acetylenic glycols, N-alkyl pyrrolidones and alkyl polyglycosides.
The Issue of Petroleum Feedstocks
For the majority of the 20th century, petroleum was used as the main feedstock for not only transportation and fuels but also chemical intermediates and co-products including surfactants. However, the heavy reliance on petroleum results in increased carbon dioxide emissions into the earth’s atmosphere, contributing to the greenhouse effect which is widely believed to lead to severe climate change throughout our planet. End-use customers are becoming more aware of this global effect and are demanding environmentally friendly products, including coatings, adhesives, sealants, and other building products for a more sustainable future. Government regulations and certifications bodies are also changing and they may force and influence the selection of feedstocks materials.
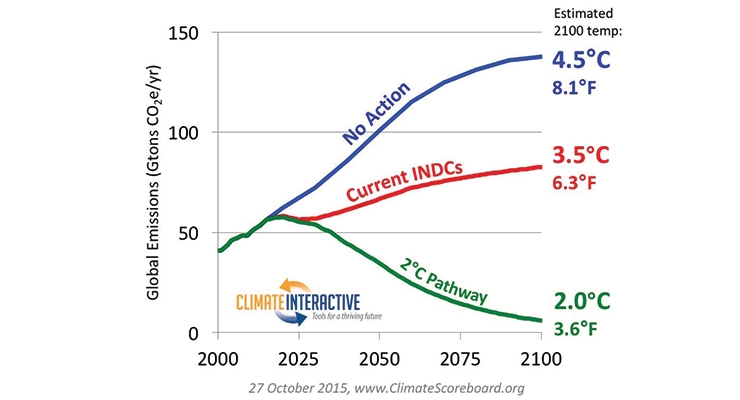
The widely recognized definition of “sustainability” is the development that meets the needs of the present without compromising the ability of future generations to meet their own needs. One of the key factors of sustainability is the mitigation of the emission of greenhouse gases and its effect on atmospheric temperature. The estimated temperature relating to global greenhouse gas emission is illustrated in Figure 4. If no action is taken, its estimated temperature will rise by 4.5 °C by 2100. If countries followed the Intended Nationally Determined Contributions (INDC), the global temperature may still go up 3.5 °C by 2100. The consensus of COP 21 (United Nations Climate Change Conference in Paris) is that the world needs to reduce carbon emissions by ca. 50% by 2050 and eliminate or significantly reduce our reliance on fossil fuels by the end of the century.
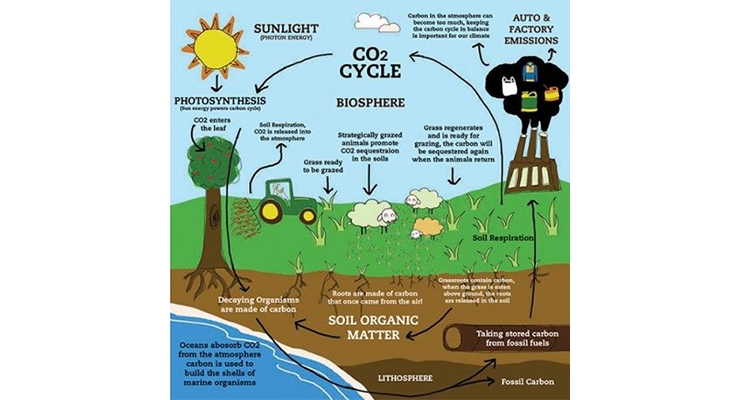
The use of fossil fuels raises serious environmental concerns. The burning of fossil fuels produces around 21.3 billion tonnes of carbon dioxide per year, but it is estimated that plants and other natural processes can only absorb about half of that amount, so there is a net increase of 10.65 billion tonnes of atmospheric carbon dioxide per year. Figure 5 shows the CO2 cycle.
One key approach to reducing the carbon footprint is to utilize renewable materials. Non-renewable materials are from resources that do not replenish in short time periods, such as minerals, metals, oil, gas or coal. Renewable materials are derived from plentiful resources that are quickly replaced by ecological cycles or agricultural processes so that the services provided by these and other linked resources are not endangered and remain available for the next generation. Therefore, bio-based products are the preferred renewable materials that will provide the sustainability for the future.
USDA BioPreferred Program
The BioPreferred Program is managed by the U.S. Department of Agriculture (USDA). Its initiative aims to assist in the development and expansion of markets for bio-based products. The program was created by the 2002 Farm Bill (legislation) and expanded as part of the 2014 Farm Bill. The increased development, purchase, and use of bio-based products will reduce our nation’s reliance on petroleum, and increase the use of renewable agricultural materials. Two major parts of the program are:
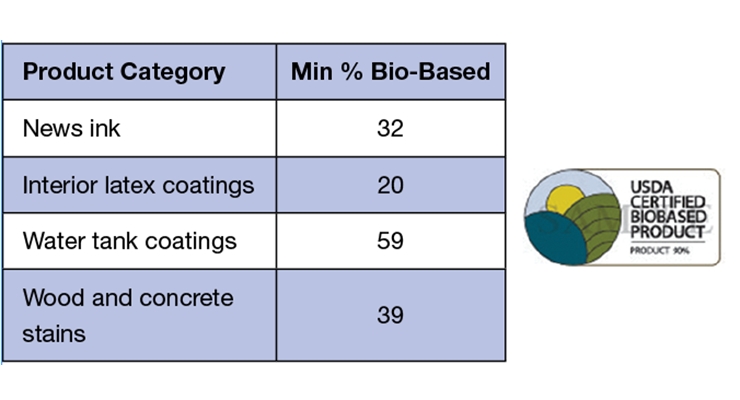
A) Mandatory purchasing requirements for federal agencies and their contractors. It establishes minimum bio-based content in 97 different categories. Figure 6 illustrates some related categories to coatings. More information can be found on the website: https://www.biopreferred.gov/BioPreferred/faces/pages/ProductCategories.xhtml
B) Issue a voluntary labeling initiative designed to provide useful information to consumers about bio-based content of the products. The program has certified more than 2,500 materials.
Renewable Surfactants
Alkyl polyglucosides are an example of a surfactant class based on renewable raw materials. These are derived from glucose, but have limited success in the marketplace, largely due to scalability and production flexibility. Other bio-based options include some betaines and proteins, but these are rarely used in the coatings market. Fermentation is used to make some production processes more environmentally friendly and bio-catalysis is also being actively researched.
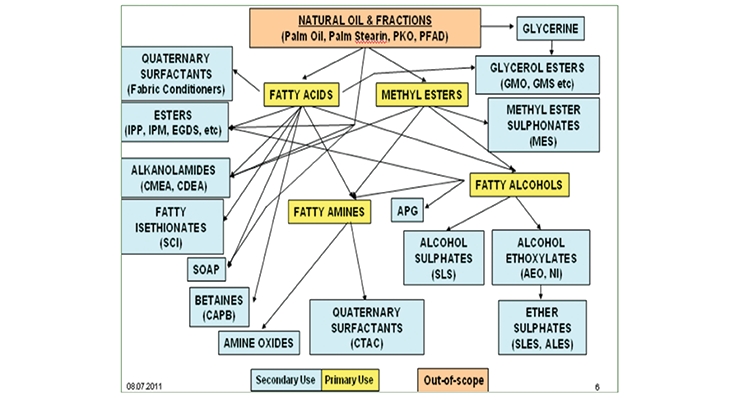
Far more abundantly available and used renewable sources are the natural oils from animal fats or plant seeds. Some of the derivatives of these oleochemicals are shown in Figure 7, which is by no means a complete representation of all possible products. The fatty content from the oils can be separated by distillation into products containing chains of 12 to 18 carbon atoms in saturated or unsaturated form. For example, lauryl, cetyl, stearyl and oleyl alcohols are commonly available and have appropriate hydrocarbon chain lengths to function as the hydrophobic tail group in surfactants. As shown in Figure 7, many renewable ionic surfactants can be made by this route including quaternary ammonium salts, amine oxides, and alcohol sulfates.
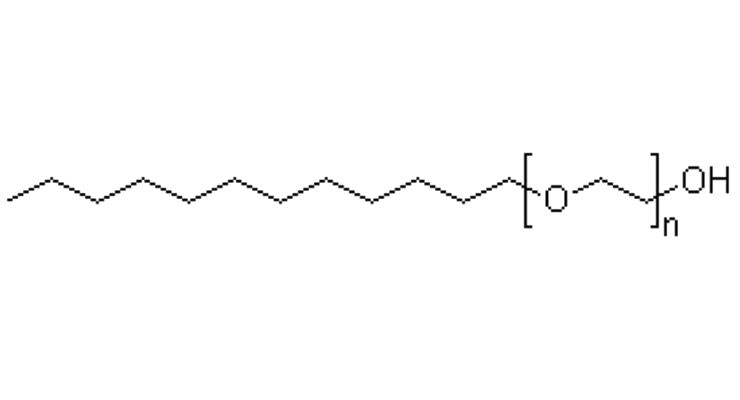
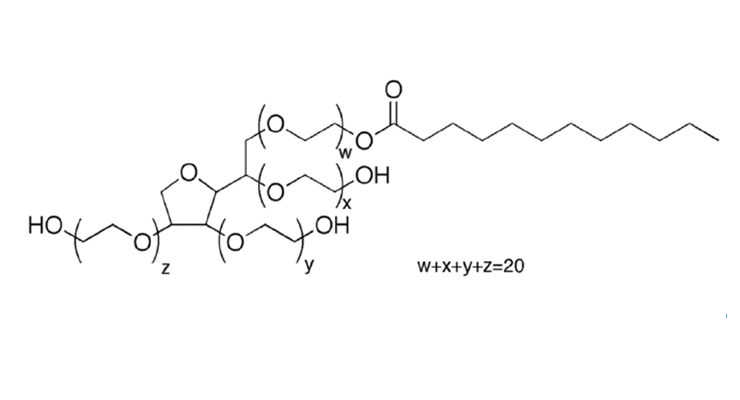
For non-ionic surfactants, however, renewability has long been limited despite the wide availability of renewable hydrophobic precursors. This is because the hydrophilic head group of most non-ionics is based on ethylene oxide (EO). Currently, EO in the United States is derived from petrochemical sources that are non-renewable. This creates a limitation, or cap, on the bio-based content of the nonionic surfactants, even where naturally sourced hydrophobes are used. For example, the bio-based carbon content of a natural lauryl (C12) alcohol with 6 moles of EO as illustrated in Figure 8 (12 bio-based carbon atoms and 12 synthetics) is only 50%. (Bio-based content is calculated using only organic carbon.)
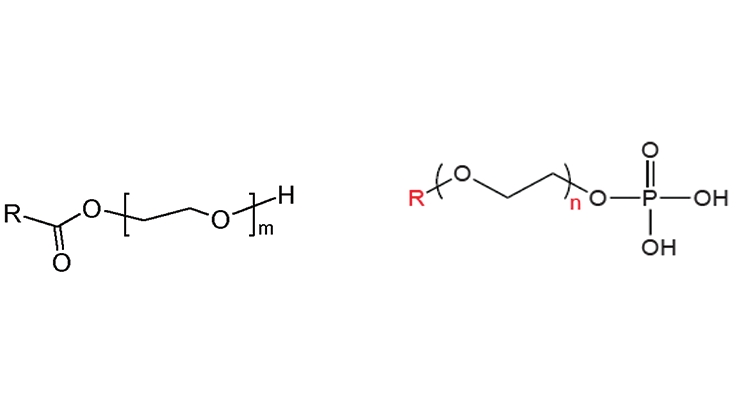
Besides fatty alcohol ethoxylates, nonionic surfactants may be based on a variety of other renewable hydrophobic fractions condensed with EO. One example is ethoxylated polysorbate (sorbitan). Usually, corn is converted into polysaccharides and then glucose. From glucose, ethanol and sorbitol (by reduction) can be made and sorbitan esters such as the one shown in Figure 9 are made by ethoxylation of the sorbitol. Controlling the degree of ethoxylation of different esters provides a wide design range for nonionic surfactants utilizing this chemistry. Other examples include ethoxylated fatty acids and phosphate esters (Figure 10), and ether sulfates among others, all limited by the same issue of EO non-renewability at the present time.
A new plant has been commissioned by Croda, Inc. in New Castle, DE to produce bio-based ethylene oxide for the conversion to nonionic surfactants. It is scheduled to start production in early 2018 and will be the first of its kind in North America (Figure 11).
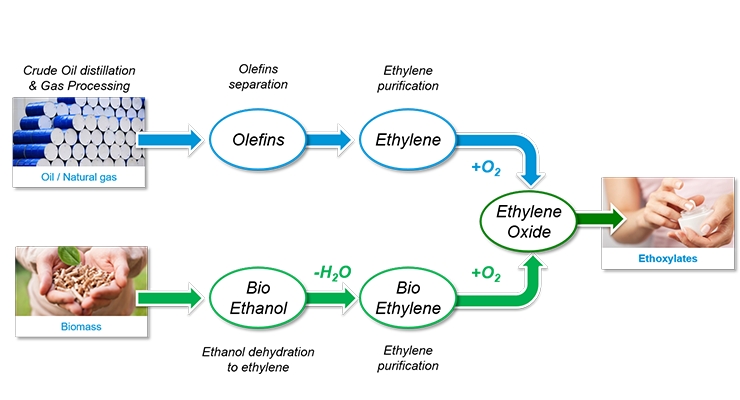
The general processes in making ethylene oxide are shown in Figure 12. Traditionally, ethylene is made from petroleum-derived olefins by a gasification process. In the newer bio-based route, naturally sourced ethanol is taken as the starting point, and converted into ethylene, which is finally transformed into ethylene oxide to be used for many surfactants.
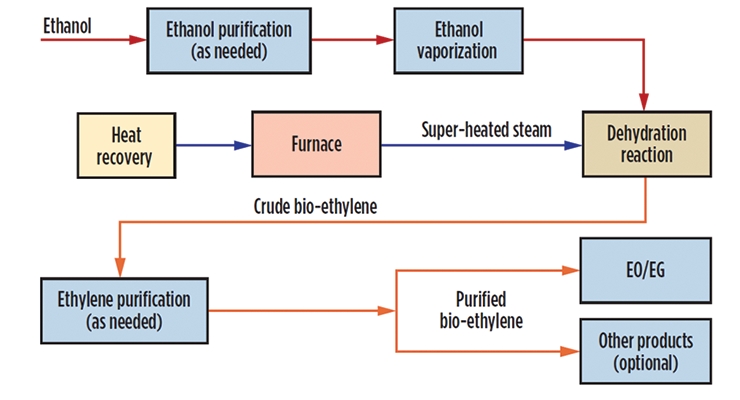
With the commission of this new plant, bio-based ethanol will be received, eliminating the transport of dangerous ethylene oxide by rail across the United States. A more complete illustration of the process beginning with bio-ethanol is shown in Figure 13. Several reactors with multiple distillation towers and hundreds of heat exchangers were put together.
With this new plant coming on stream, ethoxylation will no longer be the limitation of renewable content for surfactants or their downstream applications. The renewable content of many products will increase, for many up to 100% bio-based. It has been tested and shown that surfactants produced with bio-based EO are equivalent in performance to synthetic material but with a reduced carbon footprint. As illustrated in Figure 14, 100% renewable surfactant is attainable.
Summary
Sustainability is a growing concern among consumers in the U.S., Western Europe and many countries in Asia. To reduce the carbon footprint, renewable materials are preferred by many companies and consumers. Ethylene oxide has been the limitation on bio-based content for many surfactants. Now bio-based ethylene oxide made from bio-based ethanol will come on stream for the first time in the United States in 2018. This will remove the barrier and increase the bio-based content of many surfactants, some to 100%. The new bio-based ethoxylation capability will also enable more products and new chemistries to be BioPreferred certified in the near future.
References
1. M. J. Rosen, Surfactants and Interfacial Phenomena, (4th ed.) Hoboken, New Jersey: John Wiley & Sons, 2012
2. D. Myers, Surfactant Science and Technology, John Wiley & Sons, 2005
3. L. Maibaum, A. R. Dinner, and D. Chandler, Micelle Formation and the Hydrophobic Effect, J. Phys. Chem. B, 2004, 108 (21), pp 6778–6781
4. http://www.beautybythegeeks.com/simple-kind-to-skin-micellar-cleansing-water/
5. Hydrocarbon Magazine, Oct 2014 and Scientific Design
As consumer awareness of the products and chemicals they use rises, many chemical companies are also striving to become more environmentally responsible. The trend in the coatings industry is toward low/zero VOC products, and many innovations in coatings technology are based on sustainable or green chemistries. The market increasingly demands the use of renewable ingredients to reduce the carbon footprint, and many chemical and coating manufacturers are striving to become more environmentally responsible. This trend is driven by a number of factors, including evolving consumer perceptions and awareness regarding the products and chemicals they use, regulatory/certification bodies and programs such as USDA BioPreferred®, and the growing implementation of corporate sustainability initiatives. Bio-based ethylene oxide (EO) will meet this demand by enabling the synthesis of various ethoxylated surfactants and emulsifiers which are 100% bio-based.
Ethoxylation is a common process used to generate a range of products for emulsification and wetting, including ethoxylated alcohols, carboxylic acids and esters. While the hydrophobic portions of many of these surfactants are already naturally sourced from plant oils, only petrochemical-derived EO has been available in North America until this time. With the production of bio-based EO in the near future, ethoxylated products can now be produced from 100% bio-based content, allowing customers to choose fully renewable products without sacrificing performance. In addition, by incorporation into synthetic base materials, the bio-based content can be significantly increased, allowing formulators to meet challenging new targets.
Surfactant Fundamentals
In coatings, especially waterborne, surfactants are used as resin emulsifiers and stabilizers, pigment dispersants, grinding aids, compatibilizers, wetting agents and in defoamer and other additive packages. The word “surfactant” is the contraction of a surface active agent. Surfactants are usually organic compounds that are amphiphilic in nature, containing a lipophilic (oil like) tail and a hydrophilic (water like) head group. The general structure is illustrated in Figure 1. This unique structure enables surfactants to adsorb onto the surfaces and interfaces in a system at low concentration. The chemistry and types of surfactants are well known.1,2
When adsorbed, surfactants will lower the surface or interfacial tension. In the bulk aqueous phase, surfactants tend not to be fully soluble but rather form aggregates as micelles³ as illustrated in Figure 2. Beside spherical, other types such as cylindrical or bilayer type micelles can be formed depending on the chemistry and structure of the surfactant. The behaviors of surfactants in liquid media contribute to their great importance and tunability in applications like foaming, emulsifying, dispersing, and wetting.
Most common surfactants contain a hydrocarbon “tail” which can be linear, branched or aromatic. Other types of surfactants with tails based on siloxane and fluorocarbons are also used, but to a lesser extent in the industry. The nature of the hydrophilic head group determines the type of the surfactant: nonionic, anionic, cationic, or amphoteric, as illustrated in Figure 3. Nonionic surfactants have no charged group in the head portion of the molecule. In structures where the head carries a negative or positive charge, it is called anionic or cationic respectively. Zwitterionic is a term for surfactants with a head group consisting of two oppositely charged species.
Common anionic surfactants are sodium, potassium or ammonium salts of alkyl sulfate, sulfonate, phosphate, and sulfosuccinate.
The most common cationic surfactants are the quaternary ammonium salts, typically bromide or chloride. For zwitterionic surfactants, amines or quaternary ammonium cations make up the cationic part and the anionic portion is commonly a sulfonate or carboxylate. Alkyl betaines, and amine oxides are pH sensitive zwitterionics, displaying anionic or nonionic properties above the isoelectric point, and cationic below the isoelectric point. Sulfobetaines and sultaines are not pH sensitive.
Nonionic surfactants have several advantages over ionic types for many applications. They tend to be more compatible in a broader range of systems, which makes them generally easier to formulate with. They have much better resistance to hard water, polyvalent metallic cations, and electrolytes. Polyoxyethylenated organic compounds dominate the market of nonionic surfactants. The hydrophobic tail group could be alkylphenols, straight or branched alcohols, polyoxypropylene glycols, sorbitans or sorbitan esters, and glycol esters or fatty acids. Other organic nonionics include tertiary acetylenic glycols, N-alkyl pyrrolidones and alkyl polyglycosides.
The Issue of Petroleum Feedstocks
For the majority of the 20th century, petroleum was used as the main feedstock for not only transportation and fuels but also chemical intermediates and co-products including surfactants. However, the heavy reliance on petroleum results in increased carbon dioxide emissions into the earth’s atmosphere, contributing to the greenhouse effect which is widely believed to lead to severe climate change throughout our planet. End-use customers are becoming more aware of this global effect and are demanding environmentally friendly products, including coatings, adhesives, sealants, and other building products for a more sustainable future. Government regulations and certifications bodies are also changing and they may force and influence the selection of feedstocks materials.
The widely recognized definition of “sustainability” is the development that meets the needs of the present without compromising the ability of future generations to meet their own needs. One of the key factors of sustainability is the mitigation of the emission of greenhouse gases and its effect on atmospheric temperature. The estimated temperature relating to global greenhouse gas emission is illustrated in Figure 4. If no action is taken, its estimated temperature will rise by 4.5 °C by 2100. If countries followed the Intended Nationally Determined Contributions (INDC), the global temperature may still go up 3.5 °C by 2100. The consensus of COP 21 (United Nations Climate Change Conference in Paris) is that the world needs to reduce carbon emissions by ca. 50% by 2050 and eliminate or significantly reduce our reliance on fossil fuels by the end of the century.
The use of fossil fuels raises serious environmental concerns. The burning of fossil fuels produces around 21.3 billion tonnes of carbon dioxide per year, but it is estimated that plants and other natural processes can only absorb about half of that amount, so there is a net increase of 10.65 billion tonnes of atmospheric carbon dioxide per year. Figure 5 shows the CO2 cycle.
One key approach to reducing the carbon footprint is to utilize renewable materials. Non-renewable materials are from resources that do not replenish in short time periods, such as minerals, metals, oil, gas or coal. Renewable materials are derived from plentiful resources that are quickly replaced by ecological cycles or agricultural processes so that the services provided by these and other linked resources are not endangered and remain available for the next generation. Therefore, bio-based products are the preferred renewable materials that will provide the sustainability for the future.
USDA BioPreferred Program
The BioPreferred Program is managed by the U.S. Department of Agriculture (USDA). Its initiative aims to assist in the development and expansion of markets for bio-based products. The program was created by the 2002 Farm Bill (legislation) and expanded as part of the 2014 Farm Bill. The increased development, purchase, and use of bio-based products will reduce our nation’s reliance on petroleum, and increase the use of renewable agricultural materials. Two major parts of the program are:
A) Mandatory purchasing requirements for federal agencies and their contractors. It establishes minimum bio-based content in 97 different categories. Figure 6 illustrates some related categories to coatings. More information can be found on the website: https://www.biopreferred.gov/BioPreferred/faces/pages/ProductCategories.xhtml
B) Issue a voluntary labeling initiative designed to provide useful information to consumers about bio-based content of the products. The program has certified more than 2,500 materials.
Renewable Surfactants
Alkyl polyglucosides are an example of a surfactant class based on renewable raw materials. These are derived from glucose, but have limited success in the marketplace, largely due to scalability and production flexibility. Other bio-based options include some betaines and proteins, but these are rarely used in the coatings market. Fermentation is used to make some production processes more environmentally friendly and bio-catalysis is also being actively researched.
Far more abundantly available and used renewable sources are the natural oils from animal fats or plant seeds. Some of the derivatives of these oleochemicals are shown in Figure 7, which is by no means a complete representation of all possible products. The fatty content from the oils can be separated by distillation into products containing chains of 12 to 18 carbon atoms in saturated or unsaturated form. For example, lauryl, cetyl, stearyl and oleyl alcohols are commonly available and have appropriate hydrocarbon chain lengths to function as the hydrophobic tail group in surfactants. As shown in Figure 7, many renewable ionic surfactants can be made by this route including quaternary ammonium salts, amine oxides, and alcohol sulfates.
For non-ionic surfactants, however, renewability has long been limited despite the wide availability of renewable hydrophobic precursors. This is because the hydrophilic head group of most non-ionics is based on ethylene oxide (EO). Currently, EO in the United States is derived from petrochemical sources that are non-renewable. This creates a limitation, or cap, on the bio-based content of the nonionic surfactants, even where naturally sourced hydrophobes are used. For example, the bio-based carbon content of a natural lauryl (C12) alcohol with 6 moles of EO as illustrated in Figure 8 (12 bio-based carbon atoms and 12 synthetics) is only 50%. (Bio-based content is calculated using only organic carbon.)
Besides fatty alcohol ethoxylates, nonionic surfactants may be based on a variety of other renewable hydrophobic fractions condensed with EO. One example is ethoxylated polysorbate (sorbitan). Usually, corn is converted into polysaccharides and then glucose. From glucose, ethanol and sorbitol (by reduction) can be made and sorbitan esters such as the one shown in Figure 9 are made by ethoxylation of the sorbitol. Controlling the degree of ethoxylation of different esters provides a wide design range for nonionic surfactants utilizing this chemistry. Other examples include ethoxylated fatty acids and phosphate esters (Figure 10), and ether sulfates among others, all limited by the same issue of EO non-renewability at the present time.
A new plant has been commissioned by Croda, Inc. in New Castle, DE to produce bio-based ethylene oxide for the conversion to nonionic surfactants. It is scheduled to start production in early 2018 and will be the first of its kind in North America (Figure 11).
The general processes in making ethylene oxide are shown in Figure 12. Traditionally, ethylene is made from petroleum-derived olefins by a gasification process. In the newer bio-based route, naturally sourced ethanol is taken as the starting point, and converted into ethylene, which is finally transformed into ethylene oxide to be used for many surfactants.
With the commission of this new plant, bio-based ethanol will be received, eliminating the transport of dangerous ethylene oxide by rail across the United States. A more complete illustration of the process beginning with bio-ethanol is shown in Figure 13. Several reactors with multiple distillation towers and hundreds of heat exchangers were put together.
With this new plant coming on stream, ethoxylation will no longer be the limitation of renewable content for surfactants or their downstream applications. The renewable content of many products will increase, for many up to 100% bio-based. It has been tested and shown that surfactants produced with bio-based EO are equivalent in performance to synthetic material but with a reduced carbon footprint. As illustrated in Figure 14, 100% renewable surfactant is attainable.
Summary
Sustainability is a growing concern among consumers in the U.S., Western Europe and many countries in Asia. To reduce the carbon footprint, renewable materials are preferred by many companies and consumers. Ethylene oxide has been the limitation on bio-based content for many surfactants. Now bio-based ethylene oxide made from bio-based ethanol will come on stream for the first time in the United States in 2018. This will remove the barrier and increase the bio-based content of many surfactants, some to 100%. The new bio-based ethoxylation capability will also enable more products and new chemistries to be BioPreferred certified in the near future.
References
1. M. J. Rosen, Surfactants and Interfacial Phenomena, (4th ed.) Hoboken, New Jersey: John Wiley & Sons, 2012
2. D. Myers, Surfactant Science and Technology, John Wiley & Sons, 2005
3. L. Maibaum, A. R. Dinner, and D. Chandler, Micelle Formation and the Hydrophobic Effect, J. Phys. Chem. B, 2004, 108 (21), pp 6778–6781
4. http://www.beautybythegeeks.com/simple-kind-to-skin-micellar-cleansing-water/
5. Hydrocarbon Magazine, Oct 2014 and Scientific Design